The Materials Tetrahedron: Mastering Processing-Structure-Properties-Performance for Advanced Drug Development
This article provides a comprehensive exploration of the Materials Science Tetrahedron, a foundational paradigm defining the interdependent relationships between a material's processing, structure, properties, and performance.
The Materials Tetrahedron: Mastering Processing-Structure-Properties-Performance for Advanced Drug Development
Abstract
This article provides a comprehensive exploration of the Materials Science Tetrahedron, a foundational paradigm defining the interdependent relationships between a material's processing, structure, properties, and performance. Tailored for researchers, scientists, and drug development professionals, we dissect this framework from four critical angles: establishing its core principles, detailing methodological applications in pharmaceuticals, addressing troubleshooting and optimization challenges, and examining validation through case studies like PHA biopolymers and magnetic drug delivery systems. The content synthesizes current research and emerging trends, including the role of AI and high-throughput experimentation, to offer a actionable guide for accelerating the rational design of advanced therapeutic materials.
Deconstructing the Paradigm: A Primer on the Materials Tetrahedron and Its Core Principles
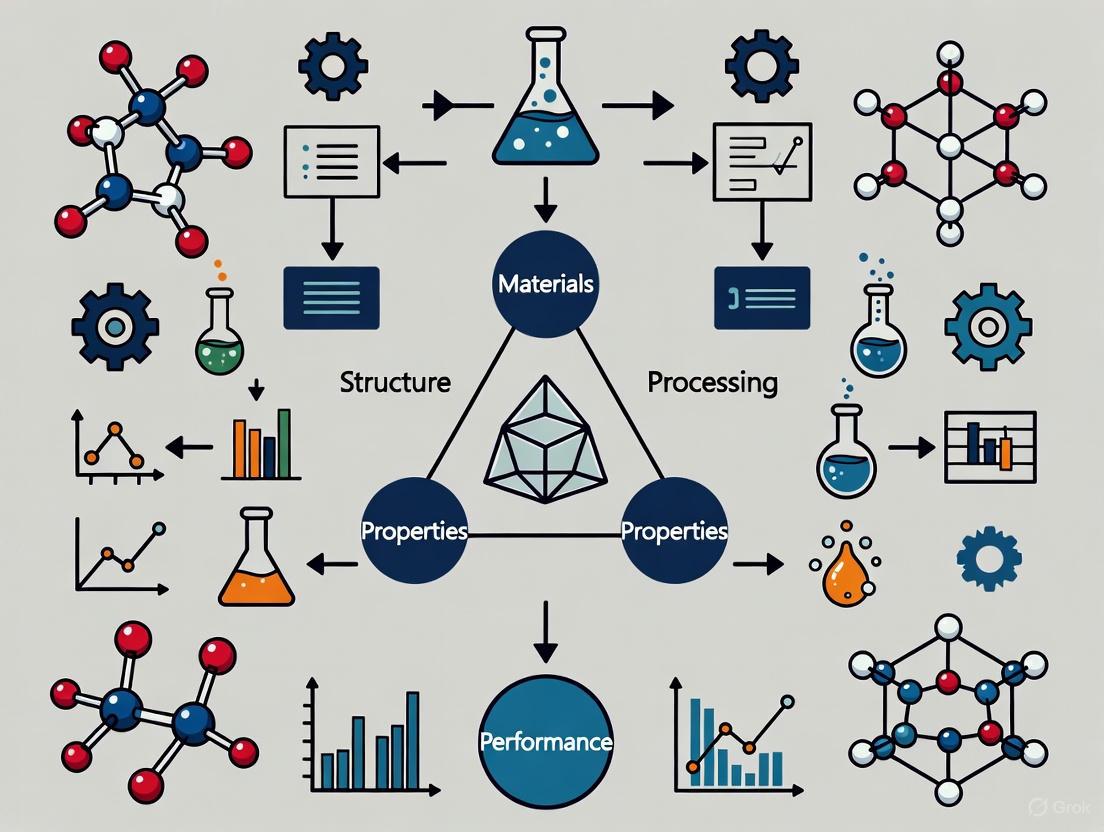
The Materials Tetrahedron is a foundational conceptual framework that captures the essence of materials science and engineering by illustrating the profound interdependence of four fundamental elements: processing, structure, properties, and performance (PSPP). This paradigm, formally introduced by the National Research Council in its 1989 report on materials science and engineering for the 1990s, has remained an enduring visual icon of the discipline for over three decades [1]. The tetrahedron's symbolic power lies in its depiction of these four elements not as a linear sequence, but as vertices of a polyhedron, with each connecting edge representing a critical, bidirectional relationship that must be understood and optimized for successful materials design [1]. The framework concisely depicts the inter-dependent relationship among the structure, properties, performance, and processing of a material, forming a scientific basis for the design and development of new materials systems [2]. In today's context of accelerated materials development, the PSPP relationship provides a crucial framework for understanding and manipulating materials behavior across diverse fields, from sustainable polymers to pharmaceutical development and national security applications.
Core Principles of the PSPP Framework
The Four Vertices: Definitions and Interrelationships
The four vertices of the materials tetrahedron form an integrated system where each element influences and is influenced by the others:
Processing refers to the methods and conditions used to synthesize, manufacture, or shape a material, including techniques such as additive manufacturing, heat treatment, casting, and chemical synthesis [3] [2]. In pharmaceuticals, this includes unit operations like milling, granulation, and compaction [2].
Structure encompasses the material's arrangement at multiple length scales, from atomic configuration and crystal structure to microstructural features, morphology, and defect architecture [4] [2]. Structure is hierarchically organized, with features at each level influencing the material's behavior.
Properties are the material's measurable responses to external stimuli, including mechanical, thermal, electrical, optical, and chemical characteristics [4] [2]. These represent what the material is capable of in terms of function.
Performance describes how the material behaves in real-world applications and service conditions, encompassing factors such as efficiency, durability, reliability, and biocompatibility [4] [5]. Performance is the ultimate criterion for materials selection.
The following table summarizes key aspects and examples for each vertex of the PSPP framework:
Table 1: Detailed Elements of the PSPP Framework Vertices
| Vertex | Key Aspects | Representative Examples |
|---|---|---|
| Processing | Synthesis methods, manufacturing techniques, heat treatment, shaping processes | Additive manufacturing, casting, milling, compaction, annealing [3] [2] |
| Structure | Atomic arrangement, crystal structure, microstructure, morphology, defects | Crystal polymorphs, grain boundaries, phase distribution, porosity [4] [2] |
| Properties | Mechanical, thermal, electrical, optical, chemical characteristics | Tensile strength, conductivity, refractive index, dissolution rate [4] [2] |
| Performance | Application behavior, service life, reliability, biocompatibility, efficacy | Drug release profile, tablet stability, component durability [4] [2] [5] |
Visualizing the Interconnections: The PSPP Workflow
The following diagram illustrates the fundamental relationships and iterative optimization cycle of the PSPP framework:
Diagram 1: PSPP Relationship Cycle
The diagram captures the fundamental PSPP workflow where processing conditions determine the material's internal structure, which governs its properties, which in turn influence its performance in practical applications. The critical feedback loop from performance back to processing enables iterative optimization of the material system.
Evolution and Modern Extensions of the Tetrahedron
The Digital Transformation: Materials-Information Twin Tetrahedra
As computational and data science methods have transformed materials research, the classic tetrahedron has been augmented with parallel concepts from information science. The Materials-Information Twin Tetrahedra (MITT) framework, inspired by the concept of a "digital twin," creates an interdependent relationship between the physical materials tetrahedron and a corresponding information tetrahedron [1]. This contemporary extension provides a holistic perspective of materials science and engineering in the presence of modern digital tools and infrastructures [1].
The information tetrahedron comprises complementary elements: methods/workflows (corresponding to processing), representations (structure), attributes (properties), and efficacy (performance), with the additional dimensions of validation and viability ensuring data quality and sustainability [1]. This dual framework incorporates FAIR data principles (Findable, Accessible, Interoperable, Reusable) and recognizes how materials systems impact and interact with other systems throughout their life cycle [1].
The TETRA Initiative: Accelerated Materials Development
The transformative potential of integrating modern computational and robotic methods with the PSPP framework is demonstrated by initiatives such as the Transforming Evaluation and Testing via Robotics and Acceleration (TETRA) program at Johns Hopkins Applied Physics Laboratory [3]. This effort reimagines the traditional tetrahedron by integrating robotics and accelerated synthesis to simultaneously explore composition and processing variants that influence properties and performance [3].
TETRA leverages advanced manufacturing techniques, including blown-powder directed energy deposition, to create hundreds of alloy variants on a single build plate, with robotic mechanical property measurement enabling rapid evaluation [3]. The ultimate vision includes creating AI "co-engineers" that work alongside human researchers, learning from materials development data to automatically recommend subsequent experiments [3].
Experimental Approaches for PSPP Investigation
Methodologies for Establishing PSPP Relationships
Establishing quantitative PSPP relationships requires carefully designed experimental and computational approaches. The following methodological framework provides a structured approach for investigating these relationships across different material systems:
Table 2: Experimental Methodologies for PSPP Relationship Analysis
| Research Focus | Experimental Methodology | Key Measurements & Characterization | Data Analysis Approaches |
|---|---|---|---|
| Processing-Structure | Combinatorial synthesis, controlled processing parameters, heat treatment variations [3] | Microscopy (SEM, TEM), diffraction (XRD), spectroscopy [6] | Microstructural quantification, statistical correlation [6] |
| Structure-Properties | Structure-property testing, in-situ characterization, property mapping [6] | Mechanical testing, thermal analysis, electrical measurements [6] [2] | Regression modeling, structure-property linkages [6] |
| Properties-Performance | Performance testing under application conditions, lifetime studies [4] [2] | Service condition simulation, accelerated aging, biocompatibility testing [4] | Failure analysis, performance prediction models [4] |
| PSP Integration | Closed-loop experimental design, multi-fidelity Bayesian optimization [6] | High-throughput characterization, autonomous data collection [3] [6] | Machine learning, Gaussian process regression [6] |
Reagent Solutions and Research Materials
The experimental investigation of PSPP relationships requires specialized materials and analytical tools. The following table outlines key research reagents and materials used in PSPP studies, particularly relevant to pharmaceutical and polymer applications:
Table 3: Essential Research Reagents and Materials for PSPP Investigations
| Reagent/Material | Function in PSPP Research | Application Examples |
|---|---|---|
| Polyhydroxyalkanoates (PHAs) | Model biodegradable polymer system for studying structure-property relationships in sustainable materials [7] [4] | Biodegradable packaging, medical implants, drug delivery systems [4] |
| Microcrystalline Cellulose | Pharmaceutical excipient for studying compaction behavior and tablet performance [2] | Tablet formulation, compactibility studies, dissolution performance [2] |
| Dual-Phase Steel Alloys | Model system for investigating microstructure-property relationships in metallic materials [6] | Mechanical property optimization, automotive components [6] |
| Polymorphic API Systems | Active pharmaceutical ingredients with multiple crystal forms for structure-property studies [2] | Solid dosage form development, bioavailability optimization [2] |
Case Studies and Applications
PSPP Framework in Pharmaceutical Development
The application of the materials tetrahedron has proven particularly valuable in pharmaceutical materials science, where it provides a scientific foundation for the design and development of drug products [2]. In this context, the tetrahedron framework has been implemented to systematize the traditionally empirical process of formulation development.
A prominent example involves the optimization of tablet compaction, where processing parameters (compression force, speed) determine the microstructure (porosity, bond formation), which governs properties (tensile strength, dissolution rate), ultimately influencing performance (drug release profile, stability) [2]. Similarly, the investigation of polymorphic systems demonstrates how crystal structure (structure) affects compaction behavior (properties) and tableting success (performance) through careful control of crystallization conditions (processing) [2].
Sustainable Biopolymers: PHA Development
The PSPP framework has been systematically applied to polyhydroxyalkanoate (PHA) biopolymers to address challenges in sustainable material development [7] [4]. This approach examines how production methods (processing) control chemical structure and morphology (structure), which determines thermal and mechanical behavior (properties), ultimately influencing biodegradation and application suitability (performance) [4].
Research has revealed that the limited chemical diversity of commercially available PHAs creates materials selection challenges, including narrow thermal processing windows and mechanical properties that are sometimes unsuitable as direct replacements for conventional plastics [4]. The PSPP framework provides a structured approach to expand the PHA design space by systematically exploring these relationships.
Computational Implementation: PROSPECT-PSPP Pipeline
The PROSPECT-PSPP computational pipeline for protein structure prediction represents a sophisticated implementation of the PSPP framework in bioinformatics [8]. This integrated system employs multiple computational tools in a coordinated workflow that mirrors the PSPP relationships: sequence preprocessing and domain identification (processing), secondary structure prediction and fold recognition (structure), model quality assessment (properties), and functional inference (performance) [8].
The pipeline demonstrates how the PSPP framework can guide the development of automated computational methodologies, with the pipeline manager controlling the flow of the prediction process by calling various tools based on results from previous steps [8].
Advanced Implementation: Workflow for Microstructure-Aware Materials Design
The following diagram illustrates an advanced, microstructure-aware closed-loop optimization framework that explicitly incorporates PSPP relationships in computational materials design:
Diagram 2: Microstructure-Aware Optimization
This workflow demonstrates the superiority of the full PSPP approach over simplified process-property (PP) relationships. Research has confirmed that explicit incorporation of microstructure knowledge in materials design frameworks significantly enhances the optimization process, proving that PSPP is superior to PP for materials design in cases where microstructure intervenes to influence properties of interest [6].
The Materials Tetrahedron, with its integrated framework of processing-structure-properties-performance relationships, continues to provide a fundamental paradigm for materials science and engineering more than three decades after its formal introduction. As the field evolves with new computational tools, characterization techniques, and data science methods, the PSPP framework has demonstrated remarkable adaptability, expanding to incorporate digital twins, artificial intelligence, and sustainable design principles.
The continued relevance of the tetrahedron lies in its ability to provide a common conceptual framework that bridges traditional materials domains—from metallic alloys to pharmaceutical systems—while enabling communication and collaboration across academia, industry, and government sectors. As materials challenges become increasingly complex and interdisciplinary, the PSPP relationship offers a structured approach for navigating the design, development, and deployment of next-generation materials systems that address critical needs in sustainability, healthcare, and national security.
In materials science and engineering, the Process-Structure-Property-Performance (PSPP) framework provides a foundational paradigm for understanding the complex relationships that govern material behavior and application suitability. This framework, often visualized as a materials tetrahedron representing the interconnected nature of these four elements, is essential for systematic materials design and development. It establishes that a material's ultimate performance in real-world applications is directly determined by its properties, which in turn emerge from its internal structure, which is controlled through specific processing techniques [9]. A comprehensive understanding of these relationships enables researchers to reverse-engineer materials, moving backward from desired performance requirements to identify the necessary structures and processing routes to achieve them.
This PSPP approach is particularly critical in advanced fields such as polymeric magnetic robotics for biomedical applications and additive manufacturing, where precise control over material behavior is necessary for functionality [9] [10]. For drug development professionals, this framework provides a structured methodology for designing biomaterials with tailored drug release profiles, biodegradation rates, and mechanical compatibility with biological tissues. This technical guide examines each component of the PSPP framework, their interrelationships, and provides experimental methodologies for their investigation, with a focus on applications relevant to advanced material systems.
Deconstructing the Four Corners: Core Concepts and Definitions
Processing
Processing encompasses the manufacturing techniques and conditions used to transform raw materials into a final form with specific structural characteristics. In advanced materials, processing parameters directly dictate the resulting hierarchical structures. Key processing techniques include:
- Additive Manufacturing: Techniques such as Selective Laser Sintering (SLS) and Direct Ink Writing (DIW) enable precise control over material architecture across multiple scales [9] [10].
- Magnetic Field-Assisted Processing: Applied during material solidification to induce directional particle assembly and enhance magnetic anisotropy in polymer composites [9].
- Thermal Processing: Controlled heating and cooling protocols that influence crystallinity, phase distribution, and structural integrity.
Critical processing parameters include temperature profiles, pressure application, magnetic field strength, and processing environment, all of which must be optimized to achieve target structures while avoiding detrimental effects such as filler demagnetization or polymer degradation [9].
Structure
Structure refers to the material's internal architecture across multiple length scales, from atomic arrangements to macroscopic features. Key structural elements include:
- Microstructure: Crystal structure, grain boundaries, phase distribution, and defects observable at microscopic scales.
- Nanoscale Features: Molecular ordering, nanoparticle dispersion, and interfacial characteristics.
- Mesoscale Architecture: Porosity, fiber orientation, and domain patterns that emerge between microscopic and macroscopic scales.
- Macroscale Geometry: Overall shape and dimensions of the finished component.
In magnetic polymer composites, structure encompasses both the polymer matrix morphology (crystalline/amorphous regions) and the spatial distribution of magnetic fillers (uniform vs. aligned configurations), which collectively determine magnetic responsiveness and mechanical behavior [9].
Properties
Properties are the measurable responses of a material to external stimuli, representing the material's capabilities and limitations. These can be categorized as:
- Physical Properties: Density, thermal conductivity, magnetic permeability
- Mechanical Properties: Elastic modulus, yield strength, fracture toughness, ductility
- Functional Properties: Shape-memory behavior, self-healing capability, drug release kinetics
- Magnetic Properties: Saturation magnetization, magnetic anisotropy, coercivity
Properties serve as the critical link between a material's internal structure and its external performance, with structure-property relationships often quantified through computational models and experimental characterization [9] [10].
Performance
Performance describes how a material system functions under actual application conditions, representing the ultimate measure of its suitability. Performance metrics are application-specific:
- Biomedical Applications: Drug delivery efficiency, targeting precision, biocompatibility, degradation profile
- Robotic Systems: Locomotion efficiency, actuation speed, operational lifetime, environmental adaptability
- Structural Components: Load-bearing capacity, durability under cyclic loading, resistance to environmental degradation
- Electronics: Charge/discharge efficiency, signal integrity, thermal management capability
For magnetic robots in drug delivery applications, key performance metrics include navigation precision through complex biological environments, controlled drug release at target sites, and minimal tissue damage during operation [9].
Quantitative PSPP Relationships in Advanced Materials
The following tables summarize key PSPP relationships in advanced material systems, highlighting how processing parameters influence structure, which subsequently determines properties and ultimate performance.
Table 1: Processing-Structure Relationships in Selective Laser Sintering of PA12
| Processing Parameter | Structural Characteristic | Quantitative Relationship | Experimental Method |
|---|---|---|---|
| Laser Power | Porosity | 62W power reduces porosity to ~2% | Micro-CT Analysis [10] |
| Scan Speed | Crystallinity | Optimal speed achieves ~30% crystallinity | DSC [10] |
| Powder Bed Temperature | Layer Adhesion | Higher temperature improves interlayer bonding | SEM Cross-section [10] |
| Magnetic Field During Curing | Particle Alignment | 500mT field creates chain-like structures | Microscopy with Image Analysis [9] |
Table 2: Structure-Property Relationships in Magnetic Polymer Composites
| Structural Feature | Material Property | Quantitative Impact | Measurement Technique |
|---|---|---|---|
| Magnetic Particle Alignment | Magnetic Anisotropy | 3-5x increase in directional response | VSM [9] |
| Porosity Distribution | Tensile Strength | 1% porosity increase reduces strength by 5-7% | Uni-axial Testing [10] |
| Interfacial Bond Quality | Fracture Toughness | Strong interface doubles energy absorption | Fracture Mechanics Tests [9] |
| Crystallinity Degree | Elastic Modulus | 10% crystallinity increase raises modulus by 15% | DMA [10] |
Table 3: Property-Performance Relationships in Medical Robotics
| Material Property | Performance Metric | Correlation | Assessment Method |
|---|---|---|---|
| Magnetic Responsiveness | Navigation Precision | Higher anisotropy enables sharper turning | Tracking in Phantoms [9] |
| Mechanical Stiffness | Locomotion Efficiency | Optimal modulus matches tissue compliance | Motion Analysis [9] |
| Biodegradation Rate | Tissue Compatibility | Controlled degradation prevents inflammation | Histology [9] |
| Surface Chemistry | Drug Loading Capacity | Functional groups increase payload by 40-60% | Spectroscopy & HPLC [9] |
Experimental Protocols for PSPP Characterization
Protocol 1: Multiscale Modeling of PSPP Relationships in Additive Manufacturing
This integrated computational framework establishes PSPP relationships for Selective Laser Sintering (SLS) of polyamide 12 (PA12) components [10].
Process Simulation
- Develop a powder-scale model simulating laser-powder interaction using optical, thermal, and geometrical properties of PA12 powder.
- Incorporate heat transfer model coupled with crystallization kinetics and densification models.
- Calculate temperature profiles, cooling rates, and sintered density with spatial resolution of 10-50 μm.
Structure Prediction
- Predict crystallinity distribution using experimentally calibrated crystallization kinetics models.
- Estimate porosity distribution from densification models with validation through micro-CT scanning.
- Construct Representative Volume Elements (RVEs) incorporating predicted porosity and crystallinity distributions.
Property Prediction
- Implement multi-mechanism constitutive model calibrated with experimental tensile tests.
- Apply periodic boundary conditions to RVEs for stress-strain response prediction.
- Calculate effective elastic modulus, yield strength, and failure strain from simulated mechanical response.
Performance Validation
- Correlate simulated mechanical properties with experimental performance metrics under application-specific loading conditions.
- Validate framework accuracy by comparing predicted and measured dimensional stability, fatigue resistance, and functional performance.
This protocol enables inverse design by establishing quantitative PSPP relationships that allow researchers to determine optimal processing parameters for desired performance outcomes [10].
Protocol 2: Experimental Characterization of Magnetic Polymer Composites
This methodology evaluates PSPP relationships in magnetically responsive polymer composites for biomedical robotics [9].
Material Processing and Fabrication
- Prepare polymer composite by homogenously dispersing magnetic particles (Fe₃O₄, NdFeB) in polymer matrix (thermoset or thermoplastic).
- Apply rotational or static magnetic fields (100-500 mT) during curing to induce particle alignment.
- Fabricate test specimens using replica molding, micro-molding, or direct-write 3D printing.
Structural Characterization
- Analyze particle distribution and alignment using scanning electron microscopy with image analysis.
- Quantify degree of alignment through orientation index calculation from microscopic images.
- Characterize polymer morphology using differential scanning calorimetry and X-ray diffraction.
Property Measurement
- Measure magnetic properties using vibrating sample magnetometry to determine saturation magnetization and coercivity.
- Characterize mechanical properties through tensile testing, dynamic mechanical analysis, and rheology.
- Evaluate functional properties such as shape-memory behavior and actuation capability under magnetic stimulation.
Performance Evaluation
- Assess actuation performance by measuring locomotion efficiency in simulated biological environments.
- Quantify targeting precision and maneuverability under controlled magnetic fields.
- Evaluate application-specific functionality (drug release profiles, diagnostic capability, therapeutic efficacy).
This comprehensive protocol enables researchers to establish quantitative correlations between processing conditions, resulting structures, emergent properties, and ultimate performance in biomedical applications [9].
Visualization of PSPP Relationships
The following diagrams illustrate key relationships and experimental workflows within the PSPP framework for advanced materials.
PSPP Framework Interrelationships
PSPP Experimental Characterization Workflow
The Scientist's Toolkit: Essential Research Materials and Reagents
Table 4: Key Research Reagents and Materials for PSPP Studies
| Material/Reagent | Function in PSPP Research | Application Examples |
|---|---|---|
| Magnetic Fillers (Fe₃O₄, NdFeB, SrFe) | Provide magnetic responsiveness | Actuation, targeting, manipulation [9] |
| Polymer Matrices (PA12, Thermosets, Hydrogels) | Structural framework | Mechanical support, biocompatibility, degradation control [9] [10] |
| Surface Modifiers (Silanes, Polymers) | Enhance particle-matrix interface | Improve dispersion, stress transfer, functionality [9] |
| Photoinitiators (Irgacure 2959, LAP) | Enable photopolymerization | 3D printing, stereolithography, patternable composites [9] |
| Rheology Modifiers (Fumed Silica, Clays) | Control viscosity and stability | Prevent sedimentation, enable 3D printing [9] |
| Crosslinking Agents (Glutaraldehyde, PEGDA) | Modify mechanical properties | Control stiffness, swelling, degradation rate [9] |
The Process-Structure-Property-Performance framework provides an essential systematic approach for advanced materials development, particularly in complex interdisciplinary fields such as biomedical robotics and additive manufacturing. By establishing quantitative relationships between these four elements, researchers can transcend traditional trial-and-error approaches and implement rational, predictive materials design [9] [10]. The continued development of multiscale modeling frameworks coupled with advanced characterization techniques will further enhance our ability to navigate this materials tetrahedron, accelerating the development of next-generation materials with tailored performance characteristics for specific biomedical and technological applications. For drug development professionals, this PSPP approach offers a structured methodology for designing carrier systems with optimized therapeutic delivery profiles, bridging the gap between materials science and pharmaceutical applications.
For over three decades, the materials tetrahedron has served as the fundamental conceptual framework of materials science and engineering, visually representing the interdependence of four core elements: processing, structure, properties, and performance [1] [11]. This symbolic polyhedron illustrates that a material's final performance is not determined by a single factor, but emerges from the complex interplay between how it is made (processing), its internal architecture across multiple length scales (structure), and its resulting characteristics (properties) [11]. The framework provides researchers with a systematic approach for rational materials design, whether moving from desired performance to required processing parameters or understanding how processing changes affect the final material behavior.
This article explores the enduring relevance of the materials tetrahedron, its modern evolution through digital twin technologies, and its practical application in developing advanced materials from metal-organic frameworks to sustainable biopolymers.
The Core Framework: Processing, Structure, Properties, Performance
The four vertices of the materials tetrahedron form a continuous cycle of cause-and-effect relationships that guide materials development:
- Processing encompasses the synthesis and manufacturing techniques used to create a material, directly influencing its internal architecture.
- Structure refers to the material's arrangement at atomic, microscopic, and macroscopic levels, including crystal structure, defects, grain boundaries, and porosity.
- Properties describe the material's characteristics and responses to external stimuli, including mechanical, electrical, thermal, and chemical behaviors.
- Performance defines how the material functions in its intended application, encompassing durability, efficiency, and reliability under operational conditions.
The framework's power lies in its bidirectional utility. The forward path (processing → structure → properties → performance) helps predict application suitability, while the inverse path (desired performance → required properties → necessary structure → appropriate processing) enables rational, target-oriented design [11].
The Digital Evolution: The Materials-Information Twin Tetrahedra
As materials research enters an era of data-intensive science, the classic tetrahedron has evolved to incorporate digital twin technology. The Materials-Information Twin Tetrahedra (MITT) framework creates an interdependent counterpart in information science, connecting physical materials research with computational approaches [1] [11].
This dual framework enables:
- Accelerated discovery through inverse design algorithms and high-throughput computational screening
- Enhanced prediction via multiphysics simulations and machine learning methods
- Improved data utility through FAIR principles (Findable, Accessible, Interoperable, Reusable) for materials data [1]
The integration of digital tools creates a virtuous cycle where materials systems generate data, information systems analyze and model this data, and the resulting insights guide further materials optimization [11].
Digital Twin Framework: The MITT connects physical materials research with information science [1] [11].
Case Study I: MOF-Based Catalysts for the Biginelli Reaction
Metal-organic frameworks (MOFs) exemplify the tetrahedron framework in designing advanced heterogeneous catalysts. Their development for the Biginelli reaction—a multicomponent process for synthesizing dihydropyrimidinone scaffolds with pharmacological importance—demonstrates precise control across all tetrahedron vertices [12].
Processing-Structure Relationships in MOF Design
MOF processing involves coordinating metal nodes with organic linkers to create crystalline porous materials. Researchers deliberately engineer structural features including:
- Ultra-high surface areas and tunable porosity for efficient reactant diffusion
- Intrinsic acidic sites at metal nodes or functionalized organic linkers
- Structural flexibility through varying metal nodes and organic linkers
- Multivariate functionalization incorporating multiple metals and ligands within a single framework [12]
These structural characteristics directly address the catalytic requirements of the Biginelli reaction, which suffers from entropy decrease in the transition state during the one-pot-three-component process. MOF cavities function as nanoreactors that stabilize the transition state, reducing activation energy and accelerating reaction rates [12].
Properties-Performance Advantages in Catalysis
The tailored structures of MOF catalysts yield enhanced properties that translate to superior performance in the Biginelli reaction:
Table 1: MOF Catalyst Advantages in Biginelli Reaction
| Structural Feature | Resulting Property | Performance Advantage |
|---|---|---|
| Abundant acidic sites | Enhanced carbonyl activation | Reduced reaction time and temperature |
| Tunable pore size | Molecular sieving capability | Improved reaction selectivity |
| Crystalline framework | Defined active site geometry | Controlled reaction mechanism |
| Non-solubility | Easy catalyst separation | Reusability and reduced waste |
The performance benefits extend beyond catalytic efficiency to environmental advantages. MOFs enable replacement of traditional homogeneous acid catalysts (HCl, H₂SO₄) that present corrosion, toxicity, and regeneration challenges, aligning with green chemistry principles [12].
Experimental Protocol: MOF-Catalyzed Biginelli Reaction
Materials and Equipment:
- Benzaldehyde, ethyl acetoacetate, urea/thiourea (equimolar amounts)
- MOF catalyst (e.g., Zr-based UiO-66 or similar acid-functionalized framework)
- Ethanol solvent
- Round-bottom flask equipped with condenser
- Heating mantle with temperature control
- Centrifuge for catalyst separation
Procedure:
- Catalyst Activation: Pre-activate MOF catalyst (typically 5-10 mol%) under vacuum at 100-150°C for 2 hours to remove solvent molecules from pores.
- Reaction Setup: Charge flask with aldehyde (1 mmol), β-ketoester (1 mmol), urea/thiourea (1.2 mmol), activated MOF catalyst, and ethanol (5 mL).
- Reaction Execution: Heat mixture at 70-80°C with continuous stirring for 1-4 hours, monitoring reaction progress by TLC or GC-MS.
- Product Isolation: Centrifuge reaction mixture to separate solid catalyst. Concentrate supernatant under reduced pressure.
- Purification: Recrystallize crude product from ethanol to obtain pure DHPMs.
- Catalyst Reuse: Wash recovered MOF catalyst with ethanol, reactivate, and reuse for subsequent cycles to assess stability.
Characterization Techniques:
- MOF Catalyst: PXRD for structural integrity, BET surface area analysis, FT-IR for functional groups, SEM/TEM for morphology
- Reaction Products: NMR, MS, and melting point determination for structural confirmation
Case Study II: Polyhydroxyalkanoate (PHA) Biopolymers
The development of sustainable polyhydroxyalkanoate (PHA) biopolymers demonstrates the tetrahedron framework's application to environmentally-conscious materials design, addressing the critical need for alternatives to conventional petroleum-based plastics [13].
Processing-Structure Challenges in PHA Development
PHA processing faces significant challenges that impact structural development and commercial viability:
- Microbial Synthesis: PHAs are synthesized and accumulated by microbes under excess carbon conditions, acting as energy storage mechanisms
- Production Limitations: Current industrial production faces hurdles in feedstock availability, low productivity/yield, and complex genetic engineering requirements
- Cost Factors: High production costs ($1.81-3.20 per lb for PHB vs $0.45-0.68 per lb for polypropylene) limit widespread adoption [13]
These processing challenges constrain structural diversity, with commercially available PHAs dominated by narrow chemistry ranges, primarily polyhydroxybutyrate (PHB) and its copolymers PHBV and PHBHHx [13].
Properties-Performance Relationships in Application
The structural limitations of commercially available PHAs directly impact their properties and performance as plastic alternatives:
Table 2: PHA Biopolymer Properties and Performance
| PHA Type | Key Properties | Performance Advantages | Performance Limitations |
|---|---|---|---|
| PHB (P3HB) | High crystallinity, brittleness | Biocompatibility, non-toxic degradation | Narrow processing window, limited degradation rate |
| PHBV | Reduced crystallinity, improved toughness | Enhanced processability, tunable degradation | Higher cost, limited chemical diversity |
| PHBHHx | Increased flexibility, lower melting point | Improved mechanical properties | Scalability challenges, production complexity |
Despite current limitations, PHAs offer crucial performance advantages for a circular plastic economy, including effective degradation in aquatic and soil environments, recyclability through chemical and biological means, and generation of non-toxic degradation products [13].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for Tetrahedron-Guided Materials Design
| Reagent/Material | Function in Research | Application Examples |
|---|---|---|
| Metal Salts (e.g., ZrCl₄, Zn(NO₃)₂) | Metal node precursors for MOF synthesis | Creating secondary building units (SBUs) in framework materials [12] |
| Organic Linkers (e.g., terephthalic acid, biphenyl-dicarboxylates) | Bridging ligands for framework construction | Establishing porosity and functionalization sites in MOFs [12] |
| Microbial Strains (e.g., Cupriavidus necator) | Biological factories for polymer synthesis | Producing PHA biopolymers from renewable feedstocks [13] |
| Solvothermal Reactors | High-pressure/temperature synthesis | Crystal growth for MOFs and coordination polymers [12] |
| Fermentation Bioreactors | Controlled biological production | Scaling up PHA synthesis with optimized nutrient conditions [13] |
The materials tetrahedron remains an indispensable framework for rational materials design, providing both foundational understanding for students and a strategic roadmap for advanced research. Its evolution through digital twin technologies has enhanced its predictive power and accelerated discovery cycles. As materials challenges grow increasingly complex—from sustainable polymers to energy storage and pharmaceutical development—the tetrahedron continues to offer a systematic approach for navigating the intricate relationships between processing, structure, properties, and performance. By integrating this classical framework with modern computational tools, researchers can more effectively design the advanced materials needed to address global technological and environmental challenges.
The materials tetrahedron is a fundamental conceptual framework in materials science that visualizes the dynamic and interdependent relationship between a material's processing, its resulting structure, its observable properties, and its ultimate performance in application [7] [3]. This paradigm, while foundational to centuries of metallurgical advancement, now provides an essential lens for understanding and innovating modern pharmaceutical development. In metallurgy, this relationship has long been understood: the method of cooling and forging steel (processing) determines its crystalline microstructure (structure), which directly dictates its hardness and tensile strength (properties), and thus its suitability for a bridge or a tool (performance). This paper argues that the same foundational principle is now being applied to the complex world of drug formulation and manufacturing, enabling a new era of advanced pharmaceuticals and personalized medicine.
The migration of this framework from metals to medicines represents a significant evolution in scientific approach. Where traditional pharmaceutical development often relied on empirical, batch-based methods, the modern paradigm, informed by the materials tetrahedron, seeks to establish predictive, science-driven models. This shift is critical for addressing contemporary challenges such as the formulation of poorly soluble Active Pharmaceutical Ingredients (APIs), the creation of complex drug delivery systems, and the move toward decentralized, on-demand manufacturing. By systematically exploring the connections between how a drug product is made, its internal architecture, its measurable characteristics, and its biological effect, researchers can accelerate development and achieve more precise therapeutic outcomes.
The Materials Science Tetrahedron: A Foundational Model
Core Principles and Definitions
The four components of the materials tetrahedron form a continuous cycle of cause and effect that is central to rational materials design. The following diagram illustrates the core interrelationships of this framework:
Figure 1: The Core Interrelationships of the Materials Tetrahedron
- Processing: The set of operations and conditions used to synthesize and shape a material. In pharmaceuticals, this encompasses unit operations like hot melt extrusion, spray drying, roller compaction, and 3D printing, as well as conditions such as temperature, pressure, and shear rate [14].
- Structure: The material's internal architecture across multiple length scales, including its molecular arrangement (e.g., crystalline vs. amorphous solid-state), microstructure (e.g., particle size and shape, porosity), and macrostructure. Structure is a direct consequence of processing.
- Properties: The measurable physical and chemical characteristics resulting from the structure. These include mechanical properties (hardness, elasticity), thermal properties (melting point, glass transition), solubility, dissolution rate, and stability.
- Performance: The material's effectiveness in its intended application or environment. For a drug product, this translates directly to therapeutic efficacy, safety profile, shelf-life, and manufacturability at scale [7].
Historical Metallurgical Origins
The empirical understanding of the PSPP relationship is ancient. Early blacksmiths manipulated the processing of steel (e.g., heating and quenching) to achieve a harder, more durable structure (martensite), which yielded properties superior for weaponry. The TETRA program at Johns Hopkins Applied Physics Laboratory (APL) is a modern embodiment of this principle, leveraging advanced tools to accelerate this discovery loop for defense-grade metallic components [3]. Historically, developing a new alloy was a painstakingly slow process, requiring the production of large ingots, sequential testing, and numerous iterative cycles. APL's TETRA approach disrupts this by using combinatorial synthesis and additive manufacturing to create and test hundreds of material variants on a single build plate, simultaneously exploring the entire composition and processing landscape [3]. This accelerated paradigm, pioneered in metallurgy, provides a template for pharmaceutical innovation.
The Paradigm Shift to Modern Pharmaceuticals
Drivers for Adoption in Pharma
The pharmaceutical industry is increasingly adopting the materials tetrahedron framework, driven by several powerful forces:
- Supply Chain Resilience: Recent geopolitical events and pandemics have exposed vulnerabilities in global pharmaceutical supply chains, prompting a wave of onshoring and investment in new, advanced manufacturing plants. Major announcements from companies like Eli Lilly, AstraZeneca, and Johnson & Johnson represent a strategic shift toward more resilient and controllable production networks [15].
- Regulatory and Policy Support: Governments are actively supporting this transition through initiatives like the U.S. FDA's "PreCheck" program, designed to fast-track the review and approval of new domestic manufacturing facilities, acknowledging the need for a more robust supply of essential medicines [15].
- Technological Innovation: The rise of Industry 4.0 technologies is making the implementation of this framework feasible. Cyber-physical systems, digital twins, and advanced data analytics allow for the real-time monitoring and control of the PSPP relationship in a way that was previously impossible [14].
- Product Complexity: The development of complex APIs, biologics, and personalized medicines demands a more sophisticated understanding of materials than ever before. Empirical methods are insufficient for engineering sophisticated drug delivery systems, necessitating a first-principles, science-based approach.
Industry 4.0 and the Smart Factory
The concept of Industry 4.0—the integration of cyber-physical systems, the Internet of Things (IoT), and cloud computing—is transforming pharmaceutical manufacturing into a data-rich, agile endeavor. This aligns perfectly with the materials tetrahedron framework [14]. Key technologies enabling this shift include:
- Digital Twin: A virtual digital equivalent of a physical product or process. It allows for real-time simulation, process optimization, and predictive analysis without disrupting actual production, dramatically decreasing development time and increasing confidence in the system [14].
- Additive Manufacturing (3D Printing): This technology epitomizes the direct link between processing and structure. It enables the fabrication of solid oral dosage forms with complex geometries (structure) that are impossible with traditional compression, allowing for customized release profiles (performance) [14].
- Advanced Robotics and AI: As demonstrated in the TETRA program, AI and robotics can act as "co-investigators," learning from development data to recommend the next experiment or even autonomously running a self-optimizing lab [3].
Applying the Tetrahedron: From Drug Formulation to Finished Product
The Pharmaceutical Materials Tetrahedron in Practice
In pharmaceutical development, the tetrahedron framework translates directly to the journey from a raw API to a finished drug product. The following workflow provides a concrete example of how this framework is applied in the development of a solid dispersion formulation, a common technique for enhancing the solubility of poorly soluble drugs:
Figure 2: A Pharmaceutical Workflow for Solid Dispersion Development
Quantitative Analysis of Processing Parameters
The relationship between processing parameters and final product properties is quantifiable. The table below summarizes key parameters and their measurable impacts on product structure and properties, illustrating the direct cause-and-effect relationships central to the tetrahedron.
Table 1: Impact of Hot Melt Extrusion (Processing) Parameters on Drug Product Attributes
| Processing Parameter | Typical Experimental Range | Impact on Solid Dispersion Structure | Resulting Property Changes |
|---|---|---|---|
| Barrel Temperature | 100°C - 180°C | Degree of API mixing & amorphous conversion [7] | Dissolution rate, physical stability |
| Screw Speed | 50 - 500 rpm | Shear-induced molecular dispersion | API particle size, homogeneity |
| Feed Rate | 0.2 - 2.0 kg/hr | Residence time in extruder | Extent of degradation, crystallinity |
| Cooling Rate | Quench vs. Slow Cool | Glassy state vs. crystalline formation | Stability, dissolution profile |
Experimental Protocols and Methodologies
Protocol: Formulating a Solid Dispersion via Hot Melt Extrusion
This protocol provides a detailed methodology for investigating the materials tetrahedron using Hot Melt Extrusion (HME) to enhance the solubility of a poorly water-soluble API.
1. Objective: To process an API-polymer blend via HME and characterize the resulting solid dispersion to understand the relationships between processing parameters, amorphous structure formation, and the resulting dissolution performance.
2. Materials (The Scientist's Toolkit):
Table 2: Essential Research Reagents and Materials
| Item | Function / Rationale |
|---|---|
| Poorly Soluble API (e.g., Itraconazole) | Model compound to demonstrate solubility enhancement. |
| Polymer Carrier (e.g., HPMCAS, PVPVA) | Matrix former that inhibits crystallization and maintains supersaturation. |
| Plasticizer (e.g., Triethyl Citrate) | Lowers processing temperature, mitigating API thermal degradation. |
| Twin-Screw Hot Melt Extruder | Provides the necessary shear and thermal energy to create a molecularly mixed amorphous dispersion. |
| Differential Scanning Calorimeter (DSC) | Confirms the conversion from crystalline API to an amorphous state. |
| X-Ray Powder Diffractometer (XRPD) | Provides definitive evidence of the loss of crystalline structure. |
| Dissolution Testing Apparatus (USP II) | Quantifies the performance enhancement (dissolution rate and extent). |
3. Detailed Procedure:
Step 1: Pre-blending
- Weigh the API and polymer in a predetermined ratio (e.g., 20:80 w/w).
- Add plasticizer at 5-10% w/w of the polymer mass.
- Blend the mixture in a turbula mixer for 15 minutes to ensure homogeneity before extrusion.
Step 2: Hot Melt Extrusion (Processing)
- Set the extruder barrel temperature profile along multiple zones, typically ramping from a low temperature in the feeding zone (e.g., 80°C) to a higher temperature in the melting and mixing zones (e.g., 150°C). The specific temperature is dependent on the glass transition temperature (Tg) of the polymer and the melting point of the API.
- Set the screw speed to a defined value, e.g., 200 rpm, to control shear stress.
- Calibrate the feeder and initiate the extrusion process at a fixed feed rate, e.g., 0.5 kg/hr.
- Collect the extrudate as it exits the die and cool it rapidly on a chilled roller.
Step 3: Post-Processing
- Milling: Size-reduce the cooled, brittle extrudate using a centrifugal mill fitted with a 1.0 mm screen.
- Storage: Store the milled powder in a sealed container under desiccated conditions until analysis.
4. Characterization (Linking Processing to Structure and Properties):
Solid-State Characterization (Structure):
- XRPD Analysis: Place powdered sample in a holder. Scan from 5° to 40° 2θ. The absence of sharp, crystalline peaks indicates the formation of an amorphous solid dispersion.
- DSC Analysis: Load 3-5 mg of sample into a sealed pan. Run a heat scan from 25°C to 250°C at 10°C/min. The disappearance of the API's melting endotherm confirms successful amorphization.
Performance Testing (Properties -> Performance):
- Dissolution Testing: Use a USP Apparatus II (paddles). Add a sample equivalent to 50 mg of API to 900 mL of dissolution medium (e.g., 0.1N HCl or phosphate buffer pH 6.8) at 37°C ± 0.5°C. Set the paddle speed to 75 rpm. Withdraw samples at 5, 10, 15, 30, 45, and 60 minutes, filter, and analyze by HPLC/UV. Compare the dissolution profile against the unprocessed crystalline API.
Protocol: Powder X-Ray Diffraction (XRPD) for Solid-State Analysis
1. Objective: To determine the crystalline or amorphous nature of the processed material.
2. Materials:
- X-Ray Powder Diffractometer
- Sample powder from Protocol 5.1
- Standard glass or zero-background sample holder
3. Procedure:
- Gently compress the powder into the sample holder to create a flat, uniform surface.
- Mount the holder in the diffractometer.
- Set parameters: Cu Kα radiation (λ = 1.5418 Å), voltage 45 kV, current 40 mA.
- Scan continuously from 5° to 40° 2θ with a step size of 0.02° and a dwell time of 1 second per step.
- Analyze the resulting diffractogram. A halo pattern indicates an amorphous material, while sharp peaks indicate crystallinity.
The journey from metallurgy to modern pharmaceuticals, guided by the enduring principles of the materials tetrahedron, represents a profound maturation of drug development. The framework provides a systematic, predictive, and scientific foundation for understanding how processing defines structure, structure governs properties, and properties ultimately dictate therapeutic performance. The adoption of this paradigm, supercharged by Industry 4.0 technologies like digital twins and additive manufacturing, is transforming the pharmaceutical landscape. It enables the development of more complex and effective drugs, enhances supply chain resilience through advanced manufacturing, and paves the way for truly personalized medicine. As the industry continues to embrace this holistic view, the path from a novel molecule to a reliable, high-performing medicine will become faster, more efficient, and fundamentally more robust.
The materials tetrahedron is a foundational conceptual framework in materials science and engineering that visually captures the fundamental, interdependent relationships between a material's processing, its resulting structure across multiple length scales, its intrinsic and extrinsic properties, and its final performance in application [16] [1]. First formally presented by the National Research Council in 1989, this enduring model has served for over three decades as a central paradigm for the field, guiding research, development, and education by illustrating that a change in one element necessarily affects the others [1] [5]. The tetrahedron's core principle is that a material's performance in service is not an isolated outcome but the culmination of a chain of relationships: processing conditions dictate the internal structure, which governs the material's properties, which ultimately determines its performance in a specific application [16] [17]. This framework provides a systematic approach for designing new materials and for troubleshooting existing ones, making it an indispensable mental model for researchers and engineers aiming to develop materials that meet extreme or novel application demands, from sustainable polymers to mission-critical defense components [7] [3] [5].
Core Principles and Elements of the Tetrahedron
The four vertices of the materials tetrahedron represent the critical domains of knowledge required for a holistic understanding of any material system. Their definitions and interrelationships are detailed below.
- Processing: This refers to the synthesis and manufacturing methods used to create a material or component. It encompasses the specific conditions, parameters, and pathways involved in transforming raw materials or precursors into a final form. Examples include casting, forging, additive manufacturing, heat treatment, and thin-film deposition. Processing is the initial, causative factor that instigates the chain of relationships within the tetrahedron [3] [17].
- Structure: Structure describes the material's internal architecture across multiple length scales, from the atomic and molecular arrangement (e.g., crystal structure, chemical bonding) to the microscopic features (e.g., grain boundaries, phase distribution) and up to the macroscopic morphology. Structure is the direct consequence of the processing history and is the primary determinant of the material's properties [7] [9].
- Properties: Properties are the measurable attributes and responses of a material to external stimuli. These include mechanical properties (e.g., strength, ductility, hardness), thermal properties (e.g., conductivity, expansion coefficient), electrical properties (e.g., conductivity, permittivity), optical properties, and chemical properties (e.g., corrosion resistance). Properties are the manifestation of the material's structure [7] [17].
- Performance: Performance characterizes how well a material functions in its intended application or service environment. It is a measure of the material's ability to meet specific engineering and economic requirements, such as fatigue life in an aerospace component, degradation rate in a biomedical implant, or efficiency in an energy conversion device. Performance is the final outcome, governed by the material's properties [7] [5].
The power of the tetrahedron model lies in the bidirectional relationships along its edges, which can be traversed in a "cause-and-effect" manner from processing to performance or in a "goal-oriented" manner backward from desired performance to required processing [1].
Quantitative Data in Tetrahedron Research
Table 1: Prevalence of Tetrahedron-Related Data in Materials Science Literature. A study of 2,536 peer-reviewed publications quantified where different types of information are reported [18].
| Information Entity | Reported in Text | Reported in Tables |
|---|---|---|
| Material Compositions | 33.21% of compositions | 85.92% of compositions |
| Material Properties | Information primarily in text | 82% of articles |
| Processing Conditions | Mostly reported in text | Less frequent |
| Testing Conditions | Mostly reported in text | Less frequent |
| Raw Materials/Precursors | 80% of articles | Less frequent |
Table 2: Distribution of Composition Table Types. An analysis of 100 randomly selected composition tables revealed structural variations that challenge automated information extraction [18].
| Table Type | Description | Prevalence |
|---|---|---|
| MCC-CI | Multi-Cell Composition with Complete Information | 36% |
| SCC-CI | Single-Cell Composition with Complete Information | 30% |
| MCC-PI | Multi-Cell Composition with Partial Information | 24% |
| SCC-PI | Single-Cell Composition with Partial Information | 10% |
Visualizing the Tetrahedron and Its Modern Extensions
The classical materials tetrahedron provides a static view of relationships. Modern research, however, requires frameworks that incorporate dynamic data flows and the digital tools used for discovery.
The Classical Materials Tetrahedron
The following diagram represents the fundamental four-element relationship that forms the core of materials science and engineering.
The Materials-Information Twin Tetrahedra (MITT) Framework
The classic tetrahedron has been reimagined for the digital age. The Materials-Information Twin Tetrahedra (MITT) framework introduces a "digital twin" for the physical materials tetrahedron, creating a nexus between materials science and information science [1] [19]. This paradigm accounts for the data, models, and digital workflows that are now central to materials research and development. The information tetrahedron comprises parallel elements: Methods/Workflows (corresponding to Processing), Representations (corresponding to Structure), Attributes (corresponding to Properties), and Efficacy (corresponding to Performance), along with the critical dimensions of Validation and Viability guided by FAIR (Findable, Accessible, Interoperable, Reusable) data principles [1]. The MITT framework facilitates a continuous, iterative cycle where materials systems generate data, and information systems provide insights that guide the improvement of materials systems.
The Integrated Research Cycle for Materials Science
The practical application of the tetrahedron occurs within a structured research cycle. This cycle integrates the scientific method with literature review and emphasizes that research is a process for expanding the community's collective knowledge, not just an individual pursuit [16]. The following workflow diagram visualizes this iterative process.
Experimental Protocols for Establishing PSPP Relationships
Establishing robust Processing-Structure-Property-Performance (PSPP) relationships requires carefully designed experimental and computational protocols. The following methodologies are drawn from cutting-edge research.
Protocol 1: Combinatorial Synthesis and High-Throughput Testing for Metallic Alloys
This protocol, as implemented in the TETRA program, leverages advanced manufacturing and robotics to dramatically accelerate the exploration of metallic materials [3].
Combinatorial Synthesis via Directed Energy Deposition (DED):
- Objective: To rapidly fabricate a library of alloy specimens with varied chemical compositions on a single build plate.
- Method: Use a blown-powder DED system. A laser melts metal powder as it is fed into the build area, solidifying layer-by-layer. The chemical composition is varied in each specimen by controlling the feed rate of different elemental or pre-alloyed powders. This allows for hundreds of discrete alloy variations to be synthesized in a single automated build cycle [3].
Automated Heat Treatment and Forging:
- Objective: To subject the combinatorial samples to a range of thermo-mechanical processing conditions.
- Method: Transfer the build plate to custom, automated heat treatment furnaces and hot forging equipment. Program these systems to apply different time-temperature profiles and deformation paths to individual or groups of samples, thereby exploring the effect of processing history on microstructure [3].
Robotic Mechanical Property Measurement:
- Objective: To autonomously test the mechanical properties of the synthesized and processed specimens.
- Method: Employ a robotic system to grip standard-sized test specimens from the build plate and load them into a mechanical testing frame. The system conducts tests (e.g., micro-tensile, hardness) and records the resulting property data (e.g., yield strength, elongation) for each unique processing-composition variant [3].
Protocol 2: Data-Driven Modeling for Metal Additive Manufacturing
This protocol uses machine learning to establish PSP links where traditional physics-based modeling is computationally prohibitive [20].
Data Generation and Curation:
- Objective: To create a dataset linking process parameters to structural features and properties.
- Method: Collect data from a combination of controlled experiments and high-fidelity thermal-fluid simulations. Key input variables (Processing) include laser power, scan speed, and scan strategy. Key output variables include Structure (e.g., porosity, lack-of-fusion defects, grain size) and Properties (e.g., yield strength, ultimate tensile strength) [20].
Surrogate Model Development and Training:
- Objective: To build a predictive model that maps process parameters to structural and property outcomes.
- Method: Employ machine learning algorithms such as Gaussian Process Regression or Deep Neural Networks. These are trained on the curated dataset to act as fast-running "surrogate models," bypassing the need for expensive simulations or experiments for new parameter sets [20].
Model Validation and Optimization:
- Objective: To validate model predictions and use the model for process optimization.
- Method: Compare model predictions against a held-out set of experimental results. Once validated, use the model in an inverse design loop to identify the optimal process parameters (e.g., laser power and scan speed) that will minimize porosity or achieve a target tensile strength [20].
Protocol 3: Fabrication and Actuation of Magnetic Polymer Composites
This protocol outlines the synthesis and testing of polymer composites for untethered magnetic robotics, highlighting specific PSPP considerations [9].
Composite Processing and Anisotropy Programming:
- Objective: To fabricate a soft polymer composite with homogeneously dispersed or directionally assembled magnetic particles.
- Method: Mix magnetic particles (e.g., NdFeB microflakes, Fe₃O₄ nanospheres) into a thermoset precursor (e.g., uncured silicone) or a thermoplastic melt. To induce magnetic anisotropy, apply an external magnetic field during the curing or solidification process. This causes particles to align into chains, programming a directional magnetic response into the material's structure [9].
Thermal Considerations During Processing:
- Objective: To preserve the programmed magnetization during fabrication.
- Method: Carefully control processing temperatures. Temperatures above the material's glass transition (Tg) or melting (Tm) temperature can unintentionally demagnetize fillers. Ensure processing temperatures remain below the Curie temperature (T_curie) of the magnetic filler to avoid erasing the pre-programmed magnetic structure [9].
Actuation Performance Testing:
- Objective: To characterize the locomotion performance of the magnetic robot in response to external magnetic fields.
- Method: Place the fabricated robot in a testing environment (e.g., liquid, surface) and subject it to controlled, time-varying magnetic fields below 100 mT. Use high-speed cameras to track and quantify locomotion modes (e.g., rolling, crawling, swimming) which are the performance metrics directly resulting from the magnetic properties and soft structure of the composite [9].
The Scientist's Toolkit: Essential Reagents and Materials
The following table details key materials and reagents used in the experimental protocols for investigating PSPP relationships in advanced material systems.
Table 3: Key Research Reagent Solutions for Tetrahedron-Related Experiments
| Item Name | Function / Role in Experiment |
|---|---|
| Metal Alloy Powder Blends | Precursors for combinatorial synthesis via Directed Energy Deposition (DED); varying composition to explore its effect on structure and properties [3]. |
| Magnetic Fillers (NdFeB, Fe₃O₄) | Functional particles incorporated into polymer matrices to impart magnetic responsiveness, enabling the actuation of soft robots [9]. |
| Thermoset Polymer Precursors | Low-viscosity resins (e.g., silicone elastomers) that serve as the matrix for composites, allowing particle mixing and alignment before cross-linking [9]. |
| FAIR-Compliant Datasets | Curated, Findable, Accessible, Interoperable, and Reusable data from experiments/simulations; the essential "reagent" for training and validating data-driven PSP models [20] [1]. |
| Gaussian Process Regression Models | A class of non-parametric, probabilistic machine learning models used as surrogate models for predicting process-structure and structure-property relationships with uncertainty quantification [20]. |
From Theory to Therapy: Applying the PSPP Framework in Pharmaceutical R&D
Integrating the Tetrahedron into the Pharmaceutical Research Cycle
The development of a new drug product is a complex, interdisciplinary endeavor that requires a deep understanding of the Active Pharmaceutical Ingredient (API) beyond its molecular structure. The "materials tetrahedron"—a conceptual framework illustrating the interdependence of processing, structure, properties, and performance—provides a powerful paradigm for navigating this complexity [21] [11]. Within pharmaceutical sciences, this framework is pivotal for ensuring that a lead solid form possesses the requisite bioavailability, physical and chemical stability, and manufacturability for successful development and commercialization [21]. The relationship between the internal structure of a solid form, its properties, and its performance within a drug product has been specifically described within a “pharmaceutical materials science” tetrahedron [21]. This whitepaper details how the systematic integration of this tetrahedron framework into the pharmaceutical research cycle de-risks development and accelerates the creation of robust, high-quality medicines.
The Tetrahedron Framework in Pharmaceutical Development
Core Elements of the Pharmaceutical Tetrahedron
In the pharmaceutical context, the four vertices of the tetrahedron take on specific, critical meanings:
- Processing: This encompasses the synthesis and manufacturing parameters of the API, including crystallization conditions (solvent, temperature, rate), milling, drying, and subsequent formulation steps. It defines the pathway to create the final drug product.
- Structure: This refers to the solid-form landscape of the API, including its molecular conformation, crystal packing, polymorphism, and potential formation of salts, co-crystals, hydrates, or solvates. The three-dimensional crystal packing arrangement is a primary determinant of material properties [21].
- Properties: These are the physicochemical characteristics resulting from the structure, such as solubility, dissolution rate, chemical stability, hygroscopicity, flowability, and mechanical properties (e.g., hardness, compaction).
- Performance: This represents the critical quality attributes (CQAs) of the final drug product, including therapeutic efficacy (influenced by bioavailability), stability throughout its shelf life, and manufacturability at scale.
The power of the framework lies in the dynamic interrelationships between these elements; a change in one necessarily affects the others. For instance, a change in crystallization processing can lead to a different polymorphic structure, which alters the solubility (properties), ultimately impacting the drug's bioavailability (performance) [21].
The Digital Twin and Informatics-Based Risk Assessment
Modern pharmaceutical development is augmenting the classical tetrahedron with digital tools. The concept of a materials–information twin tetrahedra (MITT) has been proposed, creating a "digital twin" for the materials tetrahedron [11]. This parallel information tetrahedron manages the data, representations, and workflows that describe the physical system, enabling predictive in-silico approaches.
A key application of this informatics-based approach is the Solid Form Health Check [21]. This is a digital risk assessment workflow that compares the crystal structure of a candidate API to knowledge derived from the Cambridge Structural Database (CSD). It analyzes:
- Intramolecular geometry to identify high-energy conformations.
- Hydrogen-bond parameters to identify weak interactions.
- Donor-acceptor pairings to rank interaction likelihoods based on statistical models [21].
This analysis, which can be performed in a matter of days once a crystal structure is obtained, provides invaluable early insight into potential stability risks and influences experimental design throughout the API development process [21].
Implementation: A Integrated Workflow for Solid Form De-risking
The following workflow, combining informatics, energetic calculations, and targeted experimentation, exemplifies the tetrahedron's integration into the pharmaceutical research cycle.
Experimental Protocol: A Combined Informatics and Energetic Health Check
This methodology is designed to comprehensively understand the solid form landscape of a given API and proactively identify stability risks [21].
- Objective: To ensure the selection of a thermodynamically or kinetically stable solid form for development by evaluating known polymorphs and assessing the risk of new forms emerging.
- Materials: The API of interest (e.g., PF-06282999, a molecule with conformational flexibility and multiple hydrogen bond donors/acceptors); materials for solvent-based and solid-state screening (various solvents, milling equipment); computational resources for informatics and density functional theory (DFT) calculations [21].
Step-by-Step Procedure:
Solid Form Screening:
- Execute a comprehensive polymorph screen using techniques like solvent-mediated transformation, slurrying, vapor sorption, and cryomilling to experimentally identify as many solid forms as possible [21].
- Characterize all discovered forms (e.g., Forms 1, 2, 3, and 4) using techniques like X-ray Powder Diffraction (XRPD) and thermal analysis.
Informatics Health Check:
- Obtain crystal structures for all identified forms.
- Using the CSD Python API, perform a Health Check analysis on each structure [21].
- Compare intramolecular geometry (bond lengths, angles, torsion angles) of the API to relevant fragments in the CSD to identify high-energy conformations.
- Analyze hydrogen-bond parameters (e.g., D-H···A distances and angles) against CSD-derived distributions to identify weak or suboptimal interactions.
- Rank hydrogen bond donor-acceptor pairings based on statistical modeling of their observed frequency in the CSD [21].
Energetic Calculations:
- Perform gas-phase Density Functional Theory (DFT) calculations to determine the relative conformational energies of the molecules as they exist in the different crystal structures.
- Calculate the lattice energies of the predicted and observed crystal structures to understand their relative thermodynamic stability [21].
- Integrate these energy calculations with the informatics analysis to gauge the risk associated with each form. A high-energy conformation in a crystal structure may indicate metastability.
Data Integration and Risk Assessment:
- Synthesize the findings from the experimental screening, informatics analysis, and energetic calculations.
- Create a relative stability ranking (e.g., Form 2 is most stable, Form 1 is metastable) to guide the final solid-form nomination [21].
The workflow for this integrated de-risking strategy is outlined in the diagram below.
Quantitative Data from a Case Study: PF-06282999
A study on PF-06282999 demonstrates the quantitative output of this workflow. The table below summarizes the key findings for its four polymorphs [21].
Table 1: Solid Form Health Check and Stability Analysis for PF-06282999 Polymorphs [21]
| Form ID | Relative Stability | Informatics Health Check Summary | Energetic Analysis (DFT) | Form Nomination Risk |
|---|---|---|---|---|
| Form 2 | Most Stable | Favorable hydrogen bonding and geometry. | Lowest lattice energy. | Low Risk |
| Form 1 | Metastable | Good hydrogen bonding; intramolecular geometry within populated distributions. | Higher energy than Form 2. | Medium Risk (Metastable) |
| Form 3 | Metastable | Reasonable hydrogen bonding; geometry within database distributions. | Higher energy than Form 2. | Medium Risk (Metastable) |
| Form 4 | Least Stable | Poor hydrogen bonding; unfavorable intramolecular geometry. | Highest lattice energy; high conformational strain. | High Risk |
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of this integrated workflow requires specific computational and experimental tools.
Table 2: Research Reagent Solutions for Tetrahedron-Based Solid Form Development
| Item / Reagent | Function / Explanation | Application in Workflow |
|---|---|---|
| Cambridge Structural Database (CSD) | A database of over 1.3 million small-molecule organic crystal structures used for informatics-based risk assessment and knowledge-based analysis [21]. | Informatics Health Check |
| CSD Python API | A programming interface that allows for partial automation of the Health Check analysis against the CSD [21]. | Informatics Health Check |
| Density Functional Theory (DFT) | A computational method for electronic structure calculations used to determine lattice energies and conformational metastability of crystal structures [21]. | Energetic Calculations |
| Crystal Structure Prediction (CSP) | A resource-intensive computational method to predict possible crystal structures ranked by their relative energies, used to assess the risk of unobserved polymorphs [21]. | Energetic & Risk Analysis |
| Solid Form Screening Kits | Collections of solvents and materials for executing high-throughput experimentation to explore the polymorphic landscape (e.g., via solvent-mediated transformation, slurrying) [21]. | Experimental Screening |
The integration of the materials tetrahedron into the pharmaceutical research cycle moves solid-form development from an empirical exercise to a predictive, knowledge-driven science. By systematically exploring the PSPP relationships and leveraging modern tools like informatics health checks, computational chemistry, and the digital twin concept, researchers can de-risk the development process. This integrated approach ensures the selection of a robust solid form, safeguarding drug product performance from the API manufacturing campaign through to the patient, and ultimately accelerating the delivery of new therapies.
The development of advanced drug delivery systems represents a significant challenge in modern medicine, requiring materials that are precisely engineered for performance, safety, and biodegradability. Polyhydroxyalkanoates (PHAs), a diverse class of microbially synthesized polyesters, have emerged as promising candidates for this application. This case study examines the design of PHA-based drug delivery systems through the conceptual framework of the materials tetrahedron, which illustrates the fundamental interrelationships between processing, structure, properties, and performance [16] [5]. This framework provides a systematic approach for materials scientists and engineers to understand how manipulation at one vertex of the tetrahedron necessarily induces changes throughout the entire system.
Within this framework, processing encompasses the microbial synthesis and subsequent fabrication of PHAs into drug carriers; structure refers to the chemical composition, molecular weight, and physical morphology of the polymers; properties include mechanical behavior, degradation kinetics, and biocompatibility; and performance ultimately measures the efficacy and safety of the drug delivery system in biological environments [16]. The inherent versatility of PHAs—derived from their diverse monomer compositions and the ability to tailor their biosynthesis—makes them particularly amenable to this structured design approach, enabling researchers to systematically engineer carriers with precisely defined drug release profiles and biological interactions [22] [23].
The PHA Research Cycle for Drug Delivery Applications
The development of PHA-based drug delivery systems follows an iterative research cycle that integrates computational design, experimental validation, and performance analysis. This systematic approach accelerates the optimization of PHA materials for specific therapeutic applications.
Research Cycle for PHA Drug Delivery Systems
This research cycle emphasizes that literature review is not merely an initial step but an ongoing process throughout the research lifecycle [16]. For PHA-based drug delivery, this involves continuously monitoring emerging findings on PHA biosynthesis, nanoparticle fabrication techniques, and biological performance data to inform the refinement of hypotheses and methodologies.
Material Processing: From Microbial Synthesis to Nanoparticle Fabrication
Biosynthesis and Recovery of PHAs
PHAs are produced by various microorganisms through fermentation processes under nutrient-limited conditions with excess carbon sources. The selection of microbial strain and carbon feedstock directly influences the monomer composition and resulting material properties of the synthesized PHA [23].
Table 1: Microorganisms and PHA Types for Drug Delivery Applications
| Microorganism | PHA Type | Monomer Composition | Key Characteristics |
|---|---|---|---|
| Pseudomonas putida | PHH, PHO, PHN | Medium-chain-length (C7-C9) | Improved mechanical flexibility, lower crystallinity, tunable degradation [24] |
| Cupriavidus necator | PHB, PHBV | Short-chain-length (C4-C5) | High crystallinity, relatively brittle, slower degradation [22] [23] |
| Recombinant E. coli | SCL-PHA, MCL-PHA | Variable based on genetic modification | High yield, tailored composition [23] |
The downstream processing and purification of PHAs are critical for biomedical applications. High-purity PHA suitable for drug delivery is typically obtained through a combination of biochemical recovery and precipitation using solvents, achieving purity levels >95.4% on average [23]. Additional purification steps, such as redissolution or pretreatment, are essential to meet medical standards, particularly for reducing endotoxin levels below 0.5 endotoxin units/mL [23].
Nanoparticle Fabrication and Drug Loading
The nanoprecipitation method (also known as solvent displacement) has emerged as a highly effective technique for fabricating PHA-based nanocarriers. This section provides a detailed experimental protocol for creating curcumin-loaded PHA nanoparticles, adaptable for various therapeutic compounds [24].
Experimental Protocol: Nanoprecipitation for Curcumin-Loaded PHA Nanoparticles
Materials Required:
- PHA polymer (PHH, PHO, or PHN)
- Curcumin (active pharmaceutical ingredient)
- Acetone (organic solvent)
- Aqueous phase (deionized water or PBS)
- Magnetic stirrer and stir bars
- Amber glass vials for storage
Procedure:
- Organic Phase Preparation: Dissolve 10 mg of PHA polymer and 2 mg of curcumin in 10 mL of acetone. Stir continuously at 300 rpm until complete dissolution (approximately 30 minutes).
- Aqueous Phase Preparation: Pour 20 mL of deionized water or PBS (pH 7.4) into a beaker equipped with a magnetic stirrer.
- Nanoprecipitation: While maintaining rapid stirring (800-1000 rpm) of the aqueous phase, slowly inject the organic phase using a syringe pump at a controlled rate of 1 mL/min.
- Solvent Evaporation: Continue stirring for 3-4 hours at room temperature to allow complete evaporation of acetone.
- Collection and Storage: Collect the nanoparticle suspension and store in amber glass vials at 4°C to maintain stability [24].
Critical Process Parameters:
- Solvent-to-water ratio: 1:2 (v/v)
- Stirring speed: 800-1000 rpm
- Injection rate: 1 mL/min
- Polymer-to-drug ratio: 5:1 (w/w)
This method leverages the rapid diffusion of acetone into the aqueous phase, inducing polymer precipitation and subsequent encapsulation of the hydrophobic drug molecules within the forming nanoparticles. The high affinity of both PHA and curcumin for acetone contributes to the high encapsulation efficiency observed with this technique [24].
Structure-Property Relationships in PHA Biopolymers
The structural characteristics of PHAs—including monomer composition, side chain length, and crystallinity—directly influence their material properties and performance as drug delivery matrices.
Table 2: Structure-Property Relationships of PHA Biopolymers
| Structural Feature | Impact on Material Properties | Effect on Drug Delivery Performance |
|---|---|---|
| Short-chain-length (SCL)(e.g., PHB, PHBV) | High crystallinity, brittle behavior, higher melting temperature | Slower, more sustained release profiles; potential brittleness in nanoparticle form [23] [25] |
| Medium-chain-length (MCL)(e.g., PHH, PHO, PHN) | Lower crystallinity, elastomeric behavior, wider processing window | More flexible nanoparticles; tunable degradation rates; potentially faster drug release [24] |
| Monomer Composition | Determines thermal and mechanical properties | Allows precise tuning of drug release kinetics and carrier degradation [22] [24] |
| Copolymer Ratios | Intermediate properties between SCL and MCL PHAs | Enables optimization of release profiles and mechanical stability [22] |
The relationship between PHA composition and nanoparticle characteristics is clearly demonstrated in experimental studies. When formulated using the nanoprecipitation method, medium-chain-length PHAs (PHH, PHO, PHN) consistently produce nanoparticles with sizes ranging from 307.5 to 315 nm and encapsulation efficiencies exceeding 80% for curcumin [24]. These structural characteristics directly influence the drug release behavior and biological performance of the resulting nanocarriers.
Performance Evaluation of PHA-Based Drug Delivery Systems
In Vitro Characterization and Drug Release Profiles
Comprehensive characterization of PHA nanocarriers involves multiple analytical techniques to assess critical quality attributes. Dynamic light scattering (DLS) measurements determine particle size and polydispersity index (PDI), with values below 0.29 indicating moderate homogeneity suitable for drug delivery applications [24]. Encapsulation efficiency (EE) is quantified spectrophotometrically by measuring the concentration of unencapsulated drug in the supernatant after nanoparticle separation.
Table 3: Performance Characteristics of Curcumin-Loaded PHA Nanoparticles
| PHA Type | Encapsulation Efficiency (%) | Particle Size (nm) | Polydispersity Index (PDI) | Stability (3 months at 4°C) |
|---|---|---|---|---|
| PHH | 80.41 ± 1.25 | 307.5 ± 3.44 | 0.219 ± 1.24 | <2% drug loss, no aggregation |
| PHO | 82.47 ± 0.11 | 309.9 ± 1.55 | 0.247 ± 1.79 | <2% drug loss, no aggregation |
| PHN | 84.35 ± 0.23 | 315 ± 2.76 | 0.289 ± 1.34 | <2% drug loss, no aggregation |
In vitro release studies conducted in phosphate-buffered saline under different pH conditions (pH 5.0 and 7.4) demonstrate sustained drug release profiles from PHA nanoparticles [24]. The release kinetics are influenced by both the PHA composition and environmental pH, with generally faster release observed under acidic conditions—a particularly advantageous property for targeted cancer therapy where the tumor microenvironment is often acidic.
Biological Performance and Safety Assessment
The biological performance of PHA-based drug delivery systems is evaluated through a series of standardized assays:
Cytocompatibility Assessment:
- Test nanoparticles on normal human cell lines (e.g., astrocytes, fibroblasts)
- Evaluate cell viability using MTT or Alamar Blue assays after 24-72 hours of exposure
- PHA nanoparticles consistently demonstrate excellent biocompatibility with minimal cytotoxicity on normal cells [24]
Therapeutic Efficacy Evaluation:
- Assess antiproliferative effects on relevant cancer cell lines (e.g., glioblastoma U87 MG, colon cancer Caco-2)
- Perform dose-response studies to determine IC50 values
- Curcumin-loaded PHA nanoparticles show significant antiproliferative effects while maintaining minimal toxicity to normal cells [24]
Safety Profiling:
- Conduct skin irritation and corrosion tests using reconstructed human epidermis models (EpiDerm)
- PHA-based formulations are typically classified as non-irritant and non-corrosive [24]
- Ensure endotoxin levels meet biomedical standards (<0.5 EU/mL) [23]
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful development of PHA-based drug delivery systems requires access to specialized materials and characterization tools. The following table outlines essential research reagents and their functions in the experimental workflow.
Table 4: Essential Research Reagents for PHA Drug Delivery Development
| Reagent/Material | Function | Application Notes |
|---|---|---|
| PHA Polymers(PHB, PHBV, PHH, PHO, PHN) | Primary matrix material for drug encapsulation | Selection based on desired release profile; MCL-PHAs offer enhanced flexibility [24] |
| Therapeutic Agents(Curcumin, chemotherapeutics) | Active pharmaceutical ingredient | Hydrophobic compounds typically achieve higher encapsulation efficiency [24] |
| Organic Solvents(Acetone, chloroform) | Dissolution of polymer and drug | Must have high affinity for both polymer and drug; solvent removal is critical [24] |
| Cell Culture Models(Normal and cancer cell lines) | Biological performance assessment | Required for cytotoxicity and efficacy studies [24] |
| Characterization Tools(DLS, spectrophotometry) | Nanoparticle analysis | Size, PDI, and encapsulation efficiency quantification [24] |
Current Challenges and Future Research Directions
Despite the significant promise of PHA-based drug delivery systems, several challenges must be addressed to advance their clinical translation:
Production and Purification Challenges:
- High production costs compared to synthetic polymers [22] [23]
- Complex downstream processing requiring chemical-intensive purification [23]
- Need for endotoxin reduction to meet medical standards [23]
Technical Performance Limitations:
- Limited systematic animal studies beyond in vitro validation [22]
- Need for improved targeting capabilities to specific tissues [22]
- Optimization of solvent safety in fabrication processes [22]
Future Research Priorities:
- Development of cost-effective production methods using mixed microbial cultures and waste feedstocks [23]
- Implementation of advanced manufacturing paradigms like the TETRA framework, which integrates artificial intelligence and robotics to accelerate materials development [3]
- Enhancement of targeting capabilities through surface functionalization of PHA nanoparticles
- Comprehensive in vivo studies to validate therapeutic efficacy and safety profiles
- Exploration of novel PHA copolymers with tailored degradation kinetics
The integration of computational approaches and high-throughput experimentation, as exemplified by the TETRA initiative, holds particular promise for accelerating the development of optimized PHA-based drug delivery systems [3]. This approach enables researchers to simultaneously explore multiple composition and processing variables, dramatically reducing the time required to establish robust structure-property-performance relationships.
The design of PHA biopolymers for drug delivery applications exemplifies the power of the materials tetrahedron framework in guiding the development of advanced biomedical materials. By systematically exploring the interrelationships between processing parameters, structural characteristics, material properties, and biological performance, researchers can rationally design PHA-based drug carriers with precisely tailored functionality. The exceptional biocompatibility, tunable degradation kinetics, and demonstrated drug delivery capabilities of PHAs position these microbial polyesters as promising candidates for next-generation therapeutic delivery systems. Future research focusing on overcoming current challenges in production scalability, targeting efficiency, and clinical validation will be essential for translating the potential of PHA-based drug delivery into clinical reality.
The advancement of targeted therapeutic systems represents a paradigm shift in biomedical intervention, aiming to enhance drug efficacy while minimizing systemic side effects. Within this domain, magnetic polymer composites (MPCs) have emerged as a transformative platform, enabling precise, remote-controlled drug delivery. The development of these systems is fundamentally guided by the materials tetrahedron principle, which elucidates the critical, interdependent relationships between processing, structure, properties, and performance (PSPP) [9]. This case study dissects these PSPP relationships within the context of MPCs engineered for targeted therapy, providing a technical framework for researchers and drug development professionals. The strategic integration of magnetic responsiveness with polymeric versatility allows for the design of miniaturized devices that can be wirelessly navigated to hard-to-reach physiological regions, enabling functionalities such as targeted drug release, hyperthermia, and localized biopsy [9] [26].
Fundamental Principles of Magnetic Polymer Composites
The Materials Tetrahedron in Therapeutic MPCs
The design of MPCs for targeted therapy is an exercise in optimizing the feedback loops within the materials tetrahedron. The processing techniques (e.g., 3D printing, molding) directly dictate the internal structure of the composite, including the dispersion and alignment of magnetic fillers within the polymer matrix. This structure, in turn, determines the fundamental properties of the system—its magnetic saturation, mechanical stiffness, biodegradation rate, and drug elution profile. Ultimately, these properties coalesce to define the therapeutic performance of the device, encompassing its targeting accuracy, drug release kinetics, and biocompatibility [9]. A failure to consider any single element of this tetrahedron can compromise the entire system, for instance, if a high-temperature processing step degrades the magnetic fillers or the therapeutic payload [9].
Magnetic Components and Their Biomedical Function
The magnetic functionality in MPCs is typically imparted by fillers such as iron oxides (Fe₃O₄ or γ-Fe₂O₃), carbonyl iron, or neodymium-iron-boron (NdFeB) [26] [27]. For biomedical applications, iron oxides are often preferred due to their established biocompatibility and the ability to render them superparamagnetic at nanoscale dimensions, preventing agglomeration upon removal of the external magnetic field [26]. The primary role of these fillers is to transduce an external magnetic field into a mechanical or thermal response within the composite. This enables wireless actuation for locomotion and triggered drug release mechanisms, which can be initiated by heat generated from hysteresis losses under an alternating magnetic field or by direct mechanical deformation of a drug-loaded polymer matrix [26].
Table 1: Key Magnetic Fillers and Their Characteristics for Targeted Therapy
| Filler Material | Key Magnetic Properties | Advantages | Disadvantages | Common Therapeutic Role |
|---|---|---|---|---|
| Iron Oxides (Fe₃O₄) | Superparamagnetic (nanoscale), Ferrimagnetic (microscale) | High biocompatibility, Low toxicity, Ease of synthesis | Lower saturation magnetization than metal powders | Actuation, Imaging, Hyperthermia |
| Carbonyl Iron (Fe(CO)₅) | Soft magnetic, High saturation magnetization | Strong magnetic response, Cost-effective | Prone to oxidation, May require coating | Actuation, Mechanical force generation |
| NdFeB | Hard magnetic, High coercivity, High energy product | Strongest permanent magnetic properties | Contains rare-earth elements, Potential cytotoxicity [28] | Permanent magnetization for complex actuation |
| Strontium Ferrite (SrFe₁₂O₁₉) | Hard magnetic, High coercivity | Rare-earth-free, Chemically stable [28] | Lower saturation magnetization | Alternative to NdFeB for permanent magnets |
Processing and Structure
Fabrication Techniques for MPCs
The processing of MPCs is pivotal in defining their structural hierarchy, from the molecular to the macroscopic scale. The chosen method must achieve a uniform dispersion of magnetic fillers to ensure homogeneous magnetic response and prevent defects that could compromise mechanical integrity or drug release profiles.
Molding and Casting: This conventional approach involves mixing magnetic particles with a polymer precursor (e.g., PDMS, hydrogel prepolymer) and curing the mixture in a mold. Applying a homogeneous magnetic field during curing induces particle chains, creating anisotropic structures with enhanced magnetic torque and tailored mechanical properties along the alignment direction [29]. This method is prized for its simplicity and efficacy in producing actuators and soft robotic grippers.
Additive Manufacturing (3D Printing): Techniques like Fused Deposition Modeling (FDM) and Direct Ink Writing (DIW) allow for the creation of complex 3D geometries with programmable magnetization patterns. By precisely controlling the printing path and the orientation of magnetic particles within the nozzle via a magnetic field, researchers can encode complex actuation behaviors such as bending, twisting, and crawling into the printed structure [9] [28]. This is crucial for designing miniaturized robots that can navigate tortuous biological environments.
Solution Casting and Self-Assembly: Used for creating thin films and coatings, this process involves dispersing fillers in a polymer solution, followed by solvent evaporation. Advanced variants can create core-shell structures, such as magnetic molecularly imprinted polymers (MMIPs), where a polymeric shell with specific binding cavities for a target molecule (e.g., a drug or toxin) is grown around a magnetic core [30].
Structure-Property Relationships
The processing technique directly governs the composite's microstructure, which is the primary determinant of its properties.
- Particle Dispersion and Aggregation: A homogeneous dispersion typically yields uniform properties, whereas particle aggregation can create stress concentrators, weakening the composite mechanically. However, controlled aggregation via magnetic field-assisted processing can be harnessed to create beneficial anisotropic structures [9] [27].
- Polymer Matrix Selection: The choice of polymer dictates biocompatibility, biodegradability, and mechanical compliance. Biodegradable polymers like poly(ε-caprolactone) (PCL) and poly(butylene succinate-ran-butylene adipate) (PBSA) are gaining traction for their ability to resorb in the body, eliminating the need for surgical extraction [28]. Elastomers like PDMS provide the softness and high deformability needed for safe interaction with biological tissues [29].
- Interfacial Adhesion: The strength of the interface between the magnetic filler and the polymer matrix is critical for efficient stress transfer and mechanical performance. Surface functionalization of fillers with coupling agents like (3-aminopropyl)triethoxysilane (APTES) is a common strategy to enhance this adhesion and improve dispersion stability [30] [27].
Diagram 1: The PSPP relationship from processing to performance.
Properties and Performance
Key Functional Properties and Their Evaluation
The performance of an MPC in targeted therapy is a direct consequence of its multifunctional properties, which must be meticulously characterized.
- Magnetic Properties: Saturation magnetization (Mₛ) defines the maximum magnetic moment, influencing the force/torque achievable, while coercivity (H꜀) indicates the resistance to demagnetization. For actuation, a high Mₛ is desirable, while for hyperthermia, materials with tailored coercivity and hysteresis losses are optimal [27] [28].
- Mechanical Properties: The Young's modulus and stretchability must often match those of biological tissues (kPa to MPa range) to ensure compliance and prevent damage. Composites can be engineered to be highly stretchable (e.g., hydrogels with >600% strain) or rigid for structural support [31] [29].
- Biodegradability and Self-Healing: Biodegradable composites obviate the need for retrieval surgery. Furthermore, self-healing properties can significantly extend the functional lifetime of an implant by autonomously repairing cracks or breaches caused by the body's harsh physiological conditions [26].
Table 2: Property-Performance Relationships in Therapeutic MPCs
| Functional Property | Measurement Techniques | Influence on Therapeutic Performance |
|---|---|---|
| Magnetic Responsiveness | Vibrating Sample Magnetometry (VSM), SQUID | Determines actuation force/torque and hyperthermia efficiency. |
| Mechanical Modulus | Tensile/Compression Testing, Dynamic Mechanical Analysis (DMA) | Affects biocompatibility and tissue compliance; too stiff may cause damage. |
| Drug Loading/Release Kinetics | UV-Vis Spectroscopy, HPLC | Dictates therapeutic dosage and release profile (burst vs. sustained). |
| Biodegradation Rate | Mass Loss in Simulated Body Fluid, GPC | Determines implant lifetime and need for secondary removal surgery. |
| Self-Healing Efficiency | Mechanical recovery tests after damage | Enhances durability and longevity of the implantable device. |
Performance in Targeted Therapeutic Applications
The integration of properties enables complex performance outcomes. For instance, a soft, biodegradable MPC with high magnetic saturation can be navigated to a tumor site. Once positioned, applying an alternating magnetic field can trigger a two-pronged attack: magnetic hyperthermia to ablate the tumor cells, and thermally-induced drug release from the polymer matrix for a combined therapeutic effect [26]. Furthermore, MPCs designed as micropumps or microvalves within implantable bioMEMS can provide controlled, on-demand drug release profiles for managing chronic diseases like diabetes or neurological disorders [26].
Experimental Protocols
Synthesis of Magnetic Molecularly Imprinted Polymer (MMIP) Sorbents
This protocol details the synthesis of core-shell MMIPs for the selective capture and removal of specific target molecules, such as toxins or hormones, from biological fluids [30].
Materials:
- Magnetic Core: Pre-synthesized or commercial Fe₃O₄ nanoparticles.
- Functional Monomer: Acrylic acid, methacrylic acid.
- Cross-linker: Ethylene glycol dimethacrylate (EGDMA).
- Template Molecule: The target therapeutic molecule (e.g., a toxin, hormone).
- Initiator: Azobisisobutyronitrile (AIBN).
- Surface Modifier: (3-aminopropyl)triethoxysilane (APTES) or Chitosan (CS).
Procedure:
- Functionalization of Magnetic Nanoparticles: Disperse Fe₃O₄ nanoparticles in ethanol. Add APTES dropwise under stirring and reflux for 6 hours to introduce amine groups onto the particle surface. Recover the APTES-Fe₃O₄ nanoparticles via magnetic separation and wash thoroughly.
- Formation of Pre-polymerization Complex: Re-disperse the APTES-Fe₃O₄ nanoparticles in a solvent. Add the template molecule and functional monomer. Allow them to incubate to form a complex via hydrogen bonding or covalent interactions.
- Polymerization: Add the cross-linker (EGDMA) and initiator (AIBN) to the mixture. Purge with nitrogen to remove oxygen and initiate thermal polymerization at 60°C for 12-24 hours.
- Template Removal: Recover the resulting MMIP particles magnetically and wash extensively with a solvent (e.g., methanol/acetic acid mixture) to leach out the template molecules, leaving behind specific recognition cavities.
- Characterization: Use Transmission Electron Microscopy (TEM) to confirm core-shell morphology (e.g., ~20-77 nm size range [30]). Confirm successful imprinting via adsorption kinetics and selectivity tests against analogous molecules.
Fabrication of Magneto-Responsive Soft Actuators via Molding
This protocol describes the fabrication of soft polymeric actuators for mechanical tasks like gripping or valve control in drug delivery devices [29].
Materials:
- Polymer Matrix: PDMS base and curing agent (e.g., Sylgard 184).
- Magnetic Filler: Carbonyl iron micropowder or Fe₃O₄ microparticles.
- Solvent: Cyclohexane (optional, for viscosity adjustment).
Procedure:
- Composite Mixture Preparation: Weigh the PDMS base and add magnetic particles at a desired weight fraction (e.g., 10-30% w/w). Mix thoroughly using a centrifugal mixer or mechanical stirrer to ensure homogeneity. To reduce viscosity, a solvent like cyclohexane can be added and later evaporated.
- Degassing: Place the mixture in a vacuum desiccator to remove entrapped air bubbles.
- Molding and Alignment: Pour the degassed mixture into a mold of the desired actuator shape (e.g., a cantilever beam). To induce magnetic anisotropy, place the mold into a uniform magnetic field (generated by Helmholtz coils or permanent magnets) during the curing process.
- Curing: Cure the composite at elevated temperature (e.g., 70°C for 2 hours) while the magnetic field is applied, "freezing" the aligned particle structures in place.
- Demagnetization and Testing: Demold the cured actuator. Characterize its actuation by applying controlled magnetic fields and measuring deflection using a camera and image analysis software.
Diagram 2: MMIP synthesis workflow.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Developing Therapeutic MPCs
| Reagent / Material | Function/Description | Example Use Case |
|---|---|---|
| Iron Oxide (Fe₃O₄) Nanoparticles | Provides superparamagnetic/ferrimagnetic core for responsiveness and hyperthermia. | Core material for MMIPs and injectable therapeutic agents. |
| Carbonyl Iron (Fe(CO)₅) Microparticles | High-magnetization soft magnetic filler for strong actuation forces. | Filler for soft composite actuators and micropumps. |
| Poly(dimethylsiloxane) (PDMS) | Biocompatible, stretchable silicone elastomer matrix. | Matrix for soft robotic grippers and implantable devices. |
| Poly(ε-caprolactone) (PCL) | Biodegradable, biocompatible thermoplastic polymer. | Matrix for temporary implants and absorbable scaffolds. |
| (3-aminopropyl)triethoxysilane (APTES) | Silane coupling agent for surface functionalization. | Improves filler-matrix adhesion and enables polymer grafting. |
| Chitosan (CS) | Biocompatible, biodegradable polysaccharide with functional groups. | Coating for magnetic particles to enhance biocompatibility. |
| Ethylene Glycol Dimethacrylate (EGDMA) | Cross-linking agent for creating rigid polymer networks. | Cross-linker in molecularly imprinted polymers (MIPs). |
| Azobisisobutyronitrile (AIBN) | Thermal free-radical initiator for polymerization reactions. | Initiator for the polymerization of MIP shells. |
This case study has delineated the profound connections between the processing, structure, properties, and performance of magnetic polymer composites for targeted therapy. The path forward for this field is rich with opportunity, driven by several key trends. There is a growing emphasis on sustainability, manifested in the development of composites using biodegradable polymer matrices (PCL, PBSA) and recycled or rare-earth-free magnetic fillers like strontium ferrite, aligning with circular economy principles [28]. Furthermore, the integration of multi-material 3D printing will enable the creation of increasingly sophisticated and miniaturized therapeutic devices with locally tailored functionalities. Finally, the next generation of MPCs will likely see a deeper convergence with other stimuli-responsive systems, giving rise to "embodied intelligence" in soft robotics—where sensing, actuation, and drug release are seamlessly integrated into a single, smart material system for unprecedented autonomy in biomedical applications [9] [26] [29].
In the demanding field of drug development, where the journey from concept to clinic is fraught with high costs and high failure rates, formulating a research program with the highest chance of success is paramount. The Heilmeier Catechism provides an indispensable framework for this challenge. Originally developed by George H. Heilmeier during his tenure as director of the Defense Advanced Research Projects Agency (DARPA), this set of questions is designed to refine and select high-potential, high-impact research programs [32]. For drug development professionals, this catechism forces a critical, early evaluation of a project's core vision, feasibility, and ultimate impact, ensuring that research efforts are not only scientifically sound but also translationally relevant. This guide details how to apply this powerful tool within the context of the materials tetrahedron—processing, structure, properties, and performance—a fundamental concept for designing advanced drug delivery systems and therapeutic materials. By using this framework, researchers can de-risk projects and create compelling proposals that resonate with funding agencies like the Advanced Research Projects Agency for Health (ARPA-H), which mandates its use for program concepts [33].
The Heilmeier Catechism: Core Questions and Rationale
The Heilmeier Catechism consists of a series of probing questions that force clarity and justification at the earliest stages of project planning. These questions have been adapted and expanded by various ARPA organizations to suit their specific missions. The table below summarizes the core questions and their critical function in the context of drug development research.
Table 1: The Core Heilmeier Catechism Questions for Drug Development
| Question Number | Core Question | Drug Development Context & Rationale |
|---|---|---|
| 1 | What are you trying to do? Articulate your objectives using absolutely no jargon. [33] [34] | Forces a clear "elevator pitch" for the therapeutic goal, understandable to non-specialists (e.g., regulators, investors). |
| 2 | How is it done today, and what are the limits of current practice? [33] [35] | Documents the standard of care and its deficiencies (e.g., poor bioavailability, high toxicity, complex dosing). |
| 3 | What is new about your approach, and why do you think you can be successful at this time? [33] [36] | Compels a statement of innovation and a rationale for its potential (e.g., new target, novel material, proprietary tech). |
| 4 | Who cares? If you succeed, what difference will it make? [33] [37] | Identifies stakeholders (patients, physicians) and defines the value proposition (e.g., improved survival, better QoL). |
| 5 | What are the risks? [33] [38] | Requires a candid assessment of technical, clinical, and regulatory hurdles that could derail the program. |
| 6 | How long will the program take? [33] [39] | Establishes a high-level timeline with key milestones (e.g., IND submission, Phase 1 completion). |
| 7 | How much will the program cost? [33] [39] | Provides a realistic budget estimate for achieving the major program milestones. |
| 8 | What are the mid-term and final exams to check for success? [33] [36] | Defines quantitative, measurable Go/No-Go criteria for progression (e.g., efficacy in animal model, PK profile). |
| 9* | How will cost, accessibility, and user experience be considered to reach everyone? [33] [40] | (ARPA-H Addition) Prompts planning for equitable access, manufacturability, and patient-centric design. |
| 10* | How might this program be misperceived or misused, and how can we prevent that? [33] [40] | (ARPA-H Addition) Encourages proactive consideration of ethical, safety, and public perception risks. |
Modern adaptations of the Catechism, particularly at ARPA-H, have introduced two crucial additional questions that are highly relevant to drug development [33] [40]. These ensure that considerations of equitable access and ethical foresight are baked into the research plan from the outset, rather than being afterthoughts.
Applying the Catechism to the Materials Tetrahedron in Drug Development
The materials tetrahedron is a foundational model in materials science that illustrates the interconnectedness of a material's processing, its resulting internal structure, its observable properties, and its final performance. In drug development, this model is directly applicable to the design of nanoparticle drug carriers, biodegradable implants, and advanced biotherapeutics. The Heilmeier Catechism provides the rigorous questioning framework needed to navigate each vertex of this tetrahedron systematically.
The following workflow diagram illustrates how the Heilmeier Catechism guides the research process from initial concept through to a defined program, all within the framework of the materials tetrahedron.
Interrogating the Tetrahedron with the Catechism
To formulate a powerful research plan, each element of the tetrahedron must be rigorously defined using the Heilmeier questions.
Processing → Structure: The synthesis and manufacturing process (Processing) dictates the nanoscale architecture of the drug delivery system (Structure). A Heilmeier-style question here is: "What is the new processing method for creating the polymer-drug conjugate, and why will it yield a more uniform particle size distribution (Structure) than current methods?" The answer must be jargon-free and justify the innovation.
Structure → Properties: The nanoscale architecture (Structure) determines the critical material properties (Properties). For example, the cross-linking density of a hydrogel (Structure) controls its drug release kinetics (Properties). A relevant question is: "What are the mid-term exams to confirm that the new dendritic structure leads to the predicted 50% increase in drug loading capacity?" This establishes a quantitative success metric.
Properties → Performance: The material's properties (Properties) ultimately govern its therapeutic effect in a biological system (Performance). A key question is: "If your new material achieves sustained release over 30 days, what difference will it make for patients with chronic conditions?" This directly links a technical property to clinical impact and answers "Who cares?"
This structured interrogation ensures that the research program is built on a chain of logical, testable hypotheses that connect laboratory-scale synthesis to ultimate clinical application.
Experimental Design and Validation
A research plan formulated using the Heilmeier Catechism must be backed by robust experimental methodologies designed to answer the specific questions posed by the framework. The following diagram outlines a generalized workflow for developing and testing a novel drug delivery system, incorporating key decision points and "exams" as mandated by the Catechism.
Detailed Experimental Protocols
The following protocols provide detailed methodologies for the key experiments cited in the workflow above, designed to generate quantitative data for the "mid-term and final exams."
Protocol: Structural Characterization of Nanoparticles
- Objective: To determine the size, size distribution (polydispersity index, PDI), and surface charge (zeta potential) of a newly synthesized polymeric nanoparticle formulation. This addresses the "Structure" vertex of the tetrahedron and provides critical data for Question 8 (mid-term exams).
- Materials:
- Nanoparticle Suspension: Purified formulation in aqueous buffer.
- Instrumentation: Dynamic Light Scattering (DLS) and Electrophoretic Light Scattering (ELS) instrument.
- Disposable Folded Capillary Cells: For zeta potential measurement.
- Cuvettes: For size measurement.
- Methodology:
- Sample Preparation: Dilute the nanoparticle suspension with filtered (0.1 µm pore size) deionized water or appropriate buffer to achieve an optimal scattering intensity. Avoid over-dilution or concentration.
- Size Measurement by DLS:
- Transfer the diluted sample into a clean cuvette.
- Place the cuvette in the instrument and set the measurement temperature to 25°C with an equilibration time of 120 seconds.
- Perform a minimum of 12 measurements per sample.
- Record the Z-average diameter (mean hydrodynamic diameter) and the PDI.
- Zeta Potential Measurement by ELS:
- Load the diluted sample into a clean, folded capillary cell using a syringe, ensuring no air bubbles are present.
- Insert the cell into the instrument and set the temperature to 25°C.
- Set the voltage parameters as per the instrument's guidelines for the specific cell.
- Conduct a minimum of 10 measurements per sample, calculating the mean and standard deviation of the zeta potential.
- Success Metrics (Mid-Term Exam): The formulation passes if it meets pre-defined criteria, e.g., Z-average diameter of 100 ± 20 nm, PDI < 0.2, and zeta potential |±30| mV for colloidal stability.
Protocol: In Vitro Drug Release Kinetics
- Objective: To quantify the rate and extent of drug release from a delivery system under simulated physiological conditions. This directly evaluates the "Properties" vertex.
- Materials:
- Dialysis Method: Dialysis membranes with appropriate molecular weight cut-off (MWCO), Franz diffusion cells, receptor compartment buffer (e.g., PBS at pH 7.4), water bath or shaking incubator.
- Sample Collection: Microcentrifuge tubes, HPLC vials.
- Analytical Instrumentation: HPLC system with UV/VIS detector.
- Methodology:
- Setup: Place a known volume of the nanoparticle suspension (equivalent to a specific drug dose) into a dialysis bag. Seal the bag securely.
- Immersion: Immerse the dialysis bag in a large volume of release medium (sink conditions) in the receptor compartment of a Franz cell. Maintain the system at 37°C with constant stirring.
- Sampling: At predetermined time intervals (e.g., 0.5, 1, 2, 4, 8, 24, 48, 72 hours), withdraw a known volume of the release medium from the receptor compartment.
- Replenishment: Immediately replace the withdrawn volume with fresh, pre-warmed release medium to maintain sink conditions.
- Analysis: Analyze the drug concentration in each sample using a validated HPLC method.
- Data Analysis: Calculate the cumulative percentage of drug released and plot it against time to generate the release profile.
- Success Metrics (Mid-Term Exam): The system passes if it demonstrates the desired release profile, such as <10% burst release within 2 hours and sustained release over 14 days, matching the project's therapeutic goals.
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key reagents and materials essential for experiments in advanced drug delivery, linking them to their function within the materials tetrahedron framework.
Table 2: Key Research Reagent Solutions for Drug Delivery Development
| Reagent/Material | Function & Rationale | Tetrahedron Vertex |
|---|---|---|
| PLGA (Poly(lactic-co-glycolic acid)) | A biodegradable, biocompatible polymer used as the matrix for controlled-release micro/nanoparticles. Its degradation rate (and thus drug release) can be tuned by the lactic/glycolic acid ratio. [41] | Processing, Structure |
| DSPE-PEG (Lipid-PEG conjugate) | Used to functionalize the surface of lipid nanoparticles (LNPs) and liposomes. PEGylation provides "stealth" properties by reducing opsonization and prolonging systemic circulation time. [41] | Structure, Properties |
| Dialysis Membranes (various MWCO) | A physical barrier to separate free drug from encapsulated or bound drug in release kinetics studies and purification steps. MWCO selection is critical for accurate measurement. [41] | Properties |
| Induced Pluripotent Stem Cells (iPSCs) | Provide a human-relevant, ethically sound cell source for creating disease models (e.g., neurons, cardiomyocytes) for high-throughput compound screening and toxicity testing. [41] | Performance |
| FRONT Program Graft Precursor Tissue | An example of a complex, engineered material. The goal is to create IND-ready, graftable neocortical tissue from iPSCs to replace damaged brain areas and restore function. [41] | All Vertices |
| Multi-Cancer Early Detection (MCED) Sensors | For programs like POSEIDON, these engineered sensors (e.g., for breath/urine) are the core material whose properties (sensitivity/specificity) determine diagnostic performance. [41] | Properties, Performance |
The Heilmeier Catechism is more than a checklist; it is a rigorous intellectual discipline that compels drug developers to confront the weaknesses and assumptions in their research plans before significant resources are committed. By applying its questions systematically to the materials tetrahedron framework, researchers can construct a logically sound, defensible, and impactful pathway from a novel material's synthesis to its ultimate therapeutic performance. This integrated approach ensures that research is not only technologically innovative but also translationally viable, addressing real-world problems in health with a clear-eyed view of the risks, costs, and metrics for success. In an increasingly competitive funding landscape, mastering this framework is not just an academic exercise—it is a critical strategy for designing research that can truly transform the future of medicine.
The processing-structure-property-performance (PSPP) relationship, often visualized as the materials science tetrahedron, provides the fundamental conceptual framework for understanding how a material's synthesis conditions dictate its internal architecture, which in turn defines its measurable characteristics and ultimate real-world functionality. However, establishing quantitative, predictive models of these relationships has traditionally been a slow, costly, and experimentally intensive process. The convergence of artificial intelligence (AI) with advanced digital tools and robust data pipelines is now fundamentally reshaping this paradigm. In fields ranging from metal additive manufacturing to the development of biodegradable polymers and magnetic composites, AI is accelerating the discovery and development of new materials by transforming the PSPP tetrahedron from a conceptual model into a predictive, data-driven engine for innovation [20] [7] [9]. This evolution is critical, as conventional experimental approaches and high-fidelity physics-based simulations are often prohibitively expensive or time-consuming for the rapid parameter optimization required by industry [20]. This technical guide examines the core AI methodologies, data infrastructure requirements, and experimental protocols that are defining the future of PSPP modeling.
AI Methodologies for Deciphering PSPP Relationships
Data-driven models are being deployed to establish nonlinear mappings between different elements of the PSPP chain. These models bypass the need for explicit physical equations, instead learning the underlying relationships directly from data.
Core Machine Learning Models and Their Applications
Table 1: Machine Learning Models and Their Applications in PSPP Modeling
| AI Model Category | Specific Techniques | Exemplary Application in PSPP | Key Advantage |
|---|---|---|---|
| Supervised Regression | Gaussian Process Regression, Random Forest, Gradient Boosting, Support Vector Machine | Predicting molten pool geometry, porosity, and ultimate tensile strength from process parameters [20] [42]. | Effectively captures complex, nonlinear relationships with quantified uncertainty. |
| Classification Models | Deep Neural Networks (DNNs), Support Vector Machines | Classifying molten pool melting regimes or defect modes from process data or in-situ monitoring signals [20]. | Enables rapid quality assessment and anomaly detection. |
| Ensemble Methods | Multi-gene Genetic Programming | Predicting bead width and open porosity in laser powder bed fusion [20]. | High generalization capability and robust performance. |
| Interpretable AI | Feature Importance Analysis (e.g., from Random Forest) | Identifying that processing parameters and porosity are key predictors of mechanical properties, with cell size being critical in dense samples [42]. | Provides physical insights, validating and guiding model reasoning. |
As shown in Table 1, the selection of an AI model is guided by the specific PSPP task. For instance, Gaussian process regression is particularly valued for modeling process parameters because it provides reliable predictions and uncertainty quantification even with limited training data [20]. In one landmark application, Tapia et al. used a Gaussian process surrogate model to predict molten pool depth in laser powder bed fusion (LPBF), which was then used to select process parameters that avoid keyhole mode melting and achieve a desirable conduction mode [20].
From Prediction to Physical Insight
The ultimate goal of AI in PSPP modeling is not merely to create a "black box" predictor but to gain deeper physical understanding. Feature importance analysis is a critical tool in this endeavor. In a comprehensive study on LPBF AlSi10Mg, a data-driven multivariate model revealed that while processing parameters and porosity were significant predictors of mechanical properties across all samples, for samples with density greater than 99.5%, the size of the sub-grain cellular structure was the highest contributing feature to predicting strength and ductility [42]. This insight directs researchers to focus on controlling microstructure to tailor final properties.
Foundational Data Pipelines for AI-Driven Materials Science
The efficacy of any AI model is contingent on the quality, quantity, and accessibility of the data it is trained on. Building robust data pipelines is therefore a prerequisite for success.
The Data Challenge and Infrastructure Components
Experimental materials data is often multimodal (combining synthesis conditions, characterization results, and property measurements) and multi-institutional, creating significant management hurdles [43]. Data tends to be distributed across different labs in varying formats, sizes, and content structures. To be usable for AI, this "data lake" must be transformed into an organized and searchable resource.
Table 2: Key Components of a Materials Data Infrastructure
| Component | Function | Example/Standard |
|---|---|---|
| Storage & Transfer | Hosts and enables secure, large-scale data transfer. | Globus cloud file storage [43]. |
| Data Ingestion | Standardizes and processes raw, heterogeneous data files into a consistent format. | Custom Python scripts for different file types (XRD, mechanical test data) [43]. |
| Indexing & Aggregation | Organizes standardized data across experiments for searchability. | Custom indexing scripts that create a unified database [43]. |
| Analysis & Visualization | Provides a user-friendly interface for interacting with data. | Web-based dashboards with filtering, plotting, and API access [43]. |
| Data Principles | Guides infrastructure design to ensure long-term value. | FAIR (Findable, Accessible, Interoperable, Reusable) principles [43]. |
A case study from a multi-institutional project on thermoelectric materials highlights this integrated approach. The team developed a web-based dashboard that automatically ingests data from a Globus endpoint, processes it with custom routines, and provides a frontend for visualization and analysis. This system allows researchers to interact with and gain insights from combined datasets without needing to download and process files locally, significantly accelerating the derivation of PSPP relationships [43].
The TETRA Paradigm: Integrating AI and Robotics
A pioneering example of a fully integrated, AI-driven PSPP pipeline is the TETRA (Transforming Evaluation and Testing via Robotics and Acceleration) initiative. This paradigm reimagines the traditional, serial materials development process by combining advanced manufacturing, robotics, and AI into a closed-loop system. The workflow, illustrated in the diagram below, demonstrates this accelerated approach:
Diagram 1: The TETRA Closed-Loop Workflow
TETRA leverages combinatorial synthesis via blown-powder directed energy deposition (DED) to print hundreds of unique alloy specimens on a single build plate. These specimens are then autonomously tested by robotic systems. The resulting multimodal data feeds an AI "co-investigator" that learns from the outcomes and recommends the next set of alloys and processes to test, dramatically compressing a development cycle that traditionally takes months into a matter of days [3]. This represents the cutting edge of digital PSPP pipelines.
Experimental Protocols for AI-Ready PSPP Data Generation
The reliability of an AI model is directly tied to the quality of the data used for its training. The following section details a representative experimental methodology for generating a comprehensive, AI-ready PSPP dataset, as applied in a study on laser powder bed fusion of AlSi10Mg [42].
Process Parameter Space Design and Sample Fabrication
Objective: To systematically explore a wide range of microstructures and properties by varying key process parameters.
- Parameters Varied: Laser power (P), scan speed (v), and hatch spacing (h) were selected as the primary variables.
- Process Map Design: A wide range of P and v values were selected across two different hatch spacings to probe regimes that produce fully dense material, lack-of-fusion (LoF) porosity, and keyholing porosity.
- Energy Density Metrics: Both volumetric energy density (VED = P/(v*h*layer thickness)) and modified volumetric energy density (MVED = P^3/(v^7)) were calculated to correlate with observed outcomes.
- Sample Fabrication: Test coupons were built using a commercial PBF-LB system under an inert argon atmosphere.
Structure Characterization: Porosity and Microstructure
Objective: To quantitatively characterize the internal structure of the fabricated samples.
- Porosity Analysis via X-ray Computed Tomography (XCT):
- Instrument: A commercial XCT system.
- Scan Parameters: A voxel size of (2-3 µm)³ was used to resolve pores.
- Data Processing: Reconstructed 3D volumes were analyzed using software (e.g., Amira-Avizo). Pores with edge lengths less than three times the voxel size were filtered out to respect detection limits. Pore size, morphology, and distribution were quantified.
- Microstructural Analysis via Electron Microscopy:
- Sample Preparation: Coupons were sectioned, mounted, and polished using standard metallographic techniques.
- Grain Structure (EBSD): Electron backscatter diffraction (EBSD) was performed to quantify grain size, morphology, and crystallographic texture.
- Sub-grain Structure (SEM): Scanning electron microscopy (SEM) imaging was used to measure the size of the fine cellular sub-structure characteristic of AM alloys.
Property Measurement: Mechanical Testing
Objective: To measure the mechanical performance of the samples.
- Uniaxial Tensile Testing:
- Standard: Tests were conducted according to ASTM E8/E8M.
- Procedure: Dog-bone-shaped tensile specimens were machined from the printed coupons and pulled to failure at a specified strain rate.
- Data Recorded: Ultimate tensile strength (UTS), yield strength (YS), and elongation at break (ductility) were extracted from the stress-strain curves.
- Vickers Microhardness:
- Procedure: A microhardness indenter was used with a specified load.
- Data Recorded: Hardness values were measured at multiple locations to account for heterogeneity.
Data Integration and AI Modeling
Objective: To unify the data and train AI models to uncover multivariate PSPP relationships.
- Data Unification: All processing parameters, structural features (from XCT and SEM/EBSD), and property measurements were compiled into a single, structured database.
- Model Training: Supervised machine learning algorithms (e.g., Random Forest regression) were trained on this dataset.
- Feature Importance: The trained models were analyzed using metrics like permutation importance or Gini importance to identify which input features (e.g., laser power, pore volume, cell size) were most critical for predicting response features (e.g., yield strength).
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Materials and Equipment for PSPP Experiments
| Item | Function/Description |
|---|---|
| AlSi10Mg Gas-Atomized Powder | The feedstock material for PBF-LB; its near-eutectic composition provides good weldability [42]. |
| Laser Powder Bed Fusion System | The AM equipment that uses a laser to selectively melt powder layers in an inert atmosphere [42]. |
| X-ray Computed Tomography | A non-destructive characterization tool for 3D quantification of internal porosity and defects [42]. |
| Scanning Electron Microscope | Used for high-resolution imaging of surface topography and microstructure [42]. |
| Electron Backscatter Diffraction | An SEM-based technique for analyzing crystallographic structure, grain orientation, and phase distribution [42]. |
| Universal Testing Machine | For conducting uniaxial tensile tests to determine mechanical properties [42]. |
| Vickers Microhardness Tester | For measuring local material hardness via indentation [42]. |
The integration of digital tools and data pipelines is fundamentally evolving the role of AI in modeling PSPP relationships. AI has transitioned from a novel predictive tool to a central component of an integrated discovery framework, capable of guiding experimental design, optimizing processes in real-time, and extracting profound physical insights from complex, multimodal datasets. As exemplified by the TETRA paradigm and sophisticated data infrastructures, the future of PSPP research lies in closed-loop, autonomous systems where AI acts as a co-investigator, relentlessly probing the materials tetrahedron to accelerate the development of next-generation materials for advanced applications.
Navigating Complexities: Troubleshooting and Optimizing Material Design
Common Pitfalls in Linking Material Processing to Final Performance
The materials science tetrahedron provides a fundamental framework for understanding the complex interrelationships between processing, structure, properties, and performance of materials [2]. In pharmaceutical development, this conceptual model establishes a scientific foundation for designing and developing new drug products by systematically linking material characteristics to clinical outcomes [2]. Despite its widespread recognition as a guiding principle, researchers frequently encounter significant challenges in effectively demonstrating and leveraging these critical relationships in practice.
This technical guide examines the common pitfalls that compromise the integrity of the materials tetrahedron framework and provides detailed methodologies to strengthen the connections between material processing and final performance. By addressing these vulnerabilities in experimental design and analysis, researchers can enhance the reliability and predictive capability of their development workflows, ultimately accelerating the transformation of pharmaceutical product development from an art to a science [2].
Fundamental Concepts: The Pharmaceutical Materials Tetrahedron
In pharmaceutical materials science, the four elements of the tetrahedron form a fundamental basis for understanding and engineering new materials to meet specific therapeutic needs [2]. The performance of a pharmaceutical material represents the ultimate clinical objective and provides the rationale for developing new materials. This desired performance dictates the properties required of the material, which in turn are determined by the material's structure at various length scales. Finally, the structure is controlled through specific processing techniques, completing the interdependent cycle [2].
The materials tetrahedron emphasizes that these four elements are intrinsically connected, with changes in one element necessarily affecting the others. For instance, different crystallization processes can yield varied polymorphic forms of the same active pharmaceutical ingredient (API), leading to substantially different physicochemical properties and ultimately affecting dissolution profiles and bioavailability [2]. This interconnectedness means that research focusing on only one or two elements without considering their relationships to the complete tetrahedron provides limited value for systematic product development.
Table: Key Elements of the Pharmaceutical Materials Tetrahedron
| Element | Definition | Pharmaceutical Examples |
|---|---|---|
| Processing | Methods used to synthesize, manipulate, or fabricate the material | Milling, crystallization, spray drying, compaction, hot melt extrusion |
| Structure | Arrangement of material components at atomic, molecular, and microscopic scales | Crystal polymorph, particle size distribution, surface morphology, porosity |
| Properties | Characteristics and behaviors of the material | Solubility, dissolution rate, compaction behavior, flowability, stability |
| Performance | Effectiveness in the intended application | Bioavailability, therapeutic efficacy, manufacturability, shelf life |
Critical Pitfalls in Experimental Design
Incomplete Characterization of Structure-Property Relationships
A fundamental pitfall in linking processing to performance lies in the incomplete characterization of material structures across multiple length scales. Researchers often focus on a single structural characteristic while neglecting others that may significantly influence the final performance. For example, when evaluating API compaction behavior, investigators might characterize particle size distribution but overlook critical aspects such as particle morphology, surface roughness, or internal porosity, all of which substantially impact tablet tensile strength [2].
The percolation theory model approach has demonstrated that the mechanical properties of compacts depend not only on the primary particle characteristics but also on the spatial arrangement and connectivity of different components within the formulation [2]. This complexity necessitates comprehensive structural characterization to establish meaningful structure-property relationships. Unfortunately, economic and time constraints often lead researchers to prioritize limited characterization protocols, resulting in incomplete understanding of the structural factors governing performance.
Inadequate Processing Parameter Controls
Pharmaceutical processing often involves multiple interconnected parameters that collectively influence the resulting material structure. A common experimental design flaw involves varying one processing parameter at a time while holding others constant, which fails to capture potentially significant interaction effects. For instance, in roller compaction, simultaneous evaluation of roll pressure, roll speed, and feed screw speed is necessary to understand their combined impact on granule properties and subsequent tablet performance [2].
The TETRA (Transforming Evaluation and Testing via Robotics and Acceleration) initiative at Johns Hopkins Applied Physics Laboratory addresses this limitation through combinatorial synthesis approaches that enable simultaneous exploration of multiple processing variants [3]. This methodology expands on traditional approaches that typically study factors serially, significantly accelerating the understanding of how processing parameters influence structure and properties. The conventional serial approach not only consumes substantial time and resources but also frequently misses critical parameter interactions that dictate final product performance.
Non-Representative Performance Testing Conditions
Perhaps the most prevalent pitfall involves testing material performance under conditions that poorly represent real-world application environments. In pharmaceutical development, this manifests as over-reliance on simplified in vitro models that fail to capture critical aspects of the in vivo environment. For example, dissolution testing using media that doesn't simulate gastrointestinal fluid composition or hydrodynamics may yield misleading predictions of in vivo performance [2].
A specialized example of this disconnect appears in testing protocols for protective gloves used with antineoplastic drugs. Recent analysis of experimental protocols revealed significant heterogeneity in test conditions, with studies utilizing seven different temperatures and seventeen different contact times [44]. Furthermore, critical parameters such as glove thickness at the tested area were reported in only 16.6% of articles, while methods sensitivity was documented in just 50% [44]. This variability and incomplete reporting complicate cross-study comparisons and limit the practical applicability of research findings to real-world occupational exposure scenarios.
Quantitative Data Gaps in Experimental Reporting
Comprehensive analysis of experimental reporting in materials science reveals significant data gaps that hinder the establishment of robust processing-performance relationships. The following table summarizes common quantitative reporting deficiencies identified across pharmaceutical materials science literature.
Table: Quantitative Data Gaps in Materials Experimental Reporting
| Data Category | Reporting Deficiency | Impact on Processing-Performance Link |
|---|---|---|
| Material Characteristics | Glove thickness at tested area reported in only 16.6% of permeation studies [44] | Precludes accurate correlation between material structure and barrier properties |
| Experimental Conditions | Temperature documented in only 58.3% of articles [44] | Limits understanding of thermal influences on material behavior |
| Method Sensitivity | Detection method sensitivity reported in only 50% of studies [44] | Obscures reliability limits of experimental measurements |
| Tested Material Area | Specific tested area reported in only 29% of articles [44] | Prevents normalization and comparison of results across studies |
| Mechanical Stresses | Quantitative description of applied stresses often missing [44] | Limits translation of laboratory results to real-use conditions |
These reporting deficiencies create substantial barriers to developing predictive models that link material processing to final performance. Incomplete documentation of experimental parameters prevents researchers from reproducing findings or understanding the specific conditions under which processing-structure-property relationships hold true. The lack of standardized reporting has prompted initiatives like the SPIRIT 2025 statement, which emphasizes comprehensive protocol documentation to enhance transparency and reproducibility in experimental research [45].
Detailed Experimental Protocols
Protocol for Comprehensive Material Characterization
Objective: To fully characterize material structure across multiple length scales to establish robust structure-property relationships.
Materials and Equipment:
- Scanning electron microscope (SEM) or atomic force microscope (AFM) for surface morphology
- X-ray powder diffractometer (XRPD) for crystal structure analysis
- Mercury intrusion porosimeter or gas adsorption system for surface area and porosity
- Laser diffraction particle size analyzer
- Inverse gas chromatography (IGC) for surface energy characterization
- Nanoindentation system for mechanical properties at particle level
Procedure:
- Sample Preparation: Obtain representative samples from at least three independent processing batches. For particulate materials, use riffling or quartering to ensure sample representativeness.
- Multi-scale Structural Analysis:
- Conduct XRPD analysis to identify crystalline phase, polymorphic form, and degree of crystallinity
- Perform SEM imaging at multiple magnifications (100x to 10,000x) to characterize particle morphology and surface topography
- Determine particle size distribution using laser diffraction with appropriate dispersion media
- Measure specific surface area using BET method with nitrogen adsorption
- Characterize pore size distribution using mercury porosimetry for macropores or gas adsorption for mesopores
- Surface Energy Characterization:
- Utilize IGC to determine dispersive and specific components of surface energy at multiple surface coverages
- Calculate surface energy heterogeneity profiles
- Local Mechanical Properties:
- Perform nanoindentation on individual particles to determine hardness and reduced modulus
- Conduct at least 25 indentations per sample to obtain statistically significant data
Data Analysis:
- Correlate structural characteristics with bulk powder properties (flow, compaction)
- Use multivariate analysis to identify dominant structural factors influencing performance
- Develop quantitative structure-property relationship models using partial least squares regression
Protocol for Processing-Structure Relationship Mapping
Objective: To systematically evaluate the impact of processing parameters on material structure using combinatorial approaches.
Materials and Equipment:
- Blown-powder directed energy deposition (DED) system or high-throughput crystallization platform
- Robotic material handling system
- Custom-designed heat treatment furnaces
- Hot forging equipment for microstructure modification
- Automated mechanical property measurement system [3]
Procedure:
- Combinatorial Processing:
- Utilize DED additive manufacturing to create multiple material variants on a single build plate
- Systematically vary chemical composition across the build plate using precision powder feeding
- Design custom 3D specimens suitable for subsequent testing
- Parallel Processing:
- Implement high-throughput crystallization to screen multiple solvent systems and cooling profiles simultaneously
- Apply design of experiments (DoE) methodology to explore processing parameter space efficiently
- Include at least 3 center points to assess reproducibility
- Structure Analysis:
- Employ automated specimen preparation and characterization
- Utilize machine learning algorithms to identify patterns in structure-property relationships
- Apply materials informatics approaches to extract knowledge from high-dimensional data
Data Analysis:
- Construct processing-structure maps using response surface methodology
- Identify critical processing parameters using statistical significance testing
- Develop processing windows that yield target material structures
Research Workflow Visualization
The Scientist's Toolkit: Essential Research Reagents and Materials
Table: Key Research Reagents and Materials for Tetrahedron Studies
| Item | Function | Application Notes |
|---|---|---|
| Blown-powder DED system | Enables combinatorial synthesis of material variants with controlled composition gradients [3] | Essential for high-throughput exploration of processing-structure relationships |
| Multi-station dissolution apparatus | Measures drug release profiles under physiologically relevant conditions | Must include hydrodynamics simulating gastrointestinal environment |
| Inverse Gas Chromatography (IGC) | Characterizes surface energy heterogeneity and specific interaction potential | Critical for understanding powder flow, compaction, and compatibility |
| Nanoindentation system | Measures mechanical properties at individual particle level | Provides insight into microstructure-property relationships |
| High-throughput crystallization platform | Enables parallel screening of multiple solvent systems and processing parameters | Accelerates polymorph discovery and crystal form optimization |
| Custom mechanical stress apparatus | Applies controlled mechanical and chemical stresses to simulate real-use conditions [44] | Essential for evaluating material performance under relevant conditions |
Effectively linking material processing to final performance requires meticulous attention to experimental design, comprehensive characterization, and relevant performance testing. The common pitfalls discussed in this guide—incomplete structural characterization, inadequate processing parameter controls, and non-representative performance testing—represent significant barriers to establishing robust processing-structure-property-performance relationships.
By implementing the detailed protocols and methodologies outlined herein, researchers can strengthen their approach to materials development and overcome these vulnerabilities. The integration of combinatorial processing methods, comprehensive characterization techniques, and relevant performance evaluation creates a foundation for predictive materials design. Furthermore, adherence to standardized reporting guidelines, such as those proposed in the SPIRIT 2025 statement, enhances the reproducibility and translational potential of research findings [45].
As the field advances, emerging technologies including artificial intelligence and robotics promise to further accelerate the materials development cycle. Initiatives like the TETRA program demonstrate how integrated computational and experimental approaches can simultaneously consider every variable that impacts performance, transforming what was traditionally a painstaking and time-consuming process into one that can be accomplished in days rather than months [3]. By embracing these innovative approaches while maintaining rigorous experimental methodology, researchers can fully leverage the materials tetrahedron framework to systematically advance pharmaceutical development.
Addressing Scalability and Cost Challenges in Biopolymer Production
The transition toward a circular plastic economy is critically dependent on the advancement of biopolymer technologies. Despite their sustainable promise, biopolymers face significant commercialization hurdles, primarily centered on scalability and production costs. This whitepaper examines these challenges through the foundational framework of the materials tetrahedron, which interlinks processing, structure, properties, and performance. By analyzing current research and industrial data, this guide provides a technical roadmap for researchers and scientists to navigate these barriers. It details innovative production methodologies, presents quantitative economic analyses, and outlines experimental protocols designed to enhance the efficiency and commercial viability of biopolymer production, thereby aligning material development with the principles of sustainable design.
Biopolymers, defined as polymers derived from renewable biological sources such as plants, microorganisms, or agricultural by-products, are poised to revolutionize sectors from packaging to medical devices [46] [47]. Their appeal lies in their potential for biodegradability, a reduced carbon footprint compared to conventional plastics, and origin from renewable resources [48]. However, their path to widespread adoption is obstructed by persistent challenges in scaling production and managing costs effectively [46] [49].
A systematic approach to overcoming these challenges is provided by the materials tetrahedron framework, a cornerstone of materials science and engineering. This paradigm posits that the performance of a material is a direct consequence of its properties, which are dictated by its internal structure (e.g., crystallinity, molecular weight, morphology), which is in turn governed by the processing techniques used in its synthesis and fabrication [7] [50]. For biopolymers, challenges in processing—such as the high cost of fermentation substrates and inefficient downstream purification—directly impact the final material's structure and key properties like mechanical strength and thermal stability. These property limitations ultimately restrict their performance in demanding applications, creating a feedback loop that hinders scalability [7]. This whitepaper leverages this framework to structure the analysis of current challenges and their potential solutions.
Quantitative Analysis of Scalability and Cost
A clear understanding of the economic landscape is essential for strategic planning and research direction. The following tables summarize key quantitative data on production costs and capital investment.
Table 1: Biopolymer Production Cost & Market Analysis
| Metric | Value/Range | Context & Notes |
|---|---|---|
| PLA Production Cost | $2-3 USD/kg | More established, but still higher than conventional polymers [48]. |
| PHA Production Cost | $3-5 USD/kg | Higher costs due to complex fermentation and purification [48]. |
| PET Production Cost | $1-2 USD/kg | Benchmark for conventional fossil-fuel-based plastic [48]. |
| Packaging Industry Demand | 35% | Leading application sector for biopolymers [48]. |
| Projected Bre-even Period | 4-7 years | For a new production plant, dependent on product and market [51]. |
Table 2: Capital Expenditure (CapEx) Breakdown for a Biopolymer Plant
| Cost Component | Key Inclusions | Significance |
|---|---|---|
| Machinery Costs | Fermentation reactors, separation systems, purification units, polymerization lines, pelletizing machines. | Largest portion of total capital expenditure [47] [51]. |
| Land & Site Development | Land registration, boundary development, civil works. | Forms a substantial foundation for operations [47]. |
| Infrastructure & Utilities | Construction, electricity, water, steam systems. | Critical for operational readiness and continuous production [47]. |
Operating expenditures (OpEx) are dominated by raw materials, which can account for a significant portion of the total operating cost [47] [51]. Factors such as supply chain disruptions and inflation are expected to increase total operational costs over time [47].
Mapping Challenges onto the Materials Tetrahedron
The core challenges of scalability and cost can be deconstructed and analyzed through the interconnected facets of the materials tetrahedron, as illustrated below.
Processing-Induced Challenges
- High Fermentation Costs: The reliance on purified sugar feedstocks like glucose and sucrose for microbial fermentation (e.g., for PHA and PLA) is a primary cost driver [47] [51]. Scaling up fermentation reactors requires significant capital investment and faces challenges in maintaining optimal conditions (e.g., temperature, pH, sterility) across large volumes [46] [7].
- Downstream Processing Complexity: After fermentation, biopolymers must be separated from cellular biomass and purified. This downstream processing often involves energy-intensive steps like centrifugation, filtration, and drying, contributing significantly to both CapEx and OpEx [47] [7].
Structure-Property-Performance Interrelationships
- Structural Limitations: Challenges in controlling the structure during processing, such as achieving a consistent molecular weight distribution or desired crystallinity in Polyhydroxyalkanoates (PHA), directly impact material properties [7]. This can result in inadequate mechanical strength or thermal stability compared to conventional polymers like PET or PP [46] [48].
- Performance Gaps: These compromised properties ultimately limit the material's performance in real-world applications, restricting its use to less demanding roles and hindering market competitiveness [46] [7]. For instance, variability in the protein content of soybean-based biopolymers can lead to batch inconsistencies, affecting performance in final products [52].
Advanced Processing Methodologies and Experimental Protocols
Addressing the "Processing" vertex of the tetrahedron is key to overcoming scalability and cost barriers. The following section details cutting-edge methodologies and a specific experimental protocol.
Innovative Processing Techniques
- Integrated Microfluidic-Ultrasonic Systems: The combination of microfluidic devices with ultrasonics offers a novel pathway for synthesizing emulsion-based biopolymers. Microfluidics enables precise control over droplet size and morphology, while ultrasonics enhances mixing and reaction kinetics. This integration can lead to higher product consistency and reduced energy consumption, although challenges in scalability to industrial volumes remain an active area of research [53].
- Genetic and Metabolic Engineering: Employing genetic engineering to modify microbial strains (e.g., E. coli, C. necator) can drastically improve biopolymer yields. Strategies include engineering metabolic pathways to utilize cheaper, lignocellulosic feedstocks and to overproduce target polymers like PHA, thereby reducing substrate costs and improving process efficiency [7] [48].
- Waste Valorization and Circular Feedstocks: A prominent trend is the shift towards second-generation feedstocks, such as agricultural residues (e.g., corn stover, bagasse), forestry waste, and other lignocellulosic biomass. This not only reduces raw material costs but also contributes to a circular economy [47] [51].
Experimental Protocol: Optimizing PHA Production via Fermentation
This protocol provides a methodology for investigating the impact of processing parameters on PHA yield and structure at a laboratory scale.
Objective: To determine the effect of carbon source and fermentation pH on the yield and molecular weight of PHA produced by a bacterial strain (e.g., Cupriavidus necator).
Research Reagent Solutions:
| Reagent/Material | Function in Experiment |
|---|---|
| Mineral Salt Medium (MSM) | Provides essential nutrients (N, P, K, Mg, trace elements) for bacterial growth in the absence of a limiting nitrogen source to induce PHA production. |
| Glucose/Sucrose | A pure carbon source to establish a baseline for PHA yield and structure. |
| Hydrolyzed Lignocellulosic Biomass | A complex, low-cost carbon source to test the feasibility of waste valorization and its impact on PHA production. |
| Bacterial Inoculum (C. necator) | The production host, selected for its known high PHA accumulation capacity. |
| Chloroform & Methanol | Solvents used in the downstream extraction and purification of PHA from lyophilized cell mass. |
Methodology:
- Inoculum Preparation: Inoculate a pre-culture of C. necator in a rich medium and incubate overnight at 30°C with shaking (200 rpm).
- Fermentation Setup:
- Set up a series of 1L bioreactors containing 500 mL of MSM.
- Supplement each reactor with a different carbon source (e.g., 20 g/L glucose, 20 g/L sucrose, 20 g/L hydrolyzed lignocellulosic sugar).
- Inoculate each bioreactor with 5% (v/v) of the pre-culture.
- Maintain temperature at 30°C and dissolved oxygen above 30%. Systematically vary the pH for identical reactors (e.g., 6.5, 7.0, 7.5) while keeping other parameters constant.
- Monitoring and Harvesting: Monitor cell density (OD600) periodically. Harvest cells by centrifugation during the late stationary phase (after ~48-72 hours).
- Downstream Processing:
- Lyophilization: Freeze-dry the cell pellet to obtain a dry biomass.
- Solvent Extraction: Soak the dry biomass in chloroform for 24 hours to solubilize PHA.
- Purification: Filter the chloroform extract to remove cell debris, then precipitate the polymer by adding a 10:1 volume of cold methanol.
- Drying: Collect the precipitated PHA by filtration and air-dry to constant weight.
- Analysis:
- Gravimetric Analysis: Calculate the PHA yield (% of dry cell weight).
- Gel Permeation Chromatography (GPC): Determine the molecular weight and distribution of the extracted PHA.
- FTIR or NMR Spectroscopy: Confirm the polymer type (e.g., PHB vs. PHBV) and monomer composition.
The workflow for this experiment is summarized below.
Strategic Pathways and Future Outlook
Overcoming the scalability and cost challenges requires a multi-pronged strategy that aligns with the materials tetrahedron framework.
- Government Policies and Incentives: Regulatory frameworks like the EU's Green Deal and the U.S. BioPreferred Program provide tax benefits and R&D grants, which de-risk investment and stimulate market growth for biopolymers [47]. Extended Producer Responsibility (EPR) laws are also pushing industries toward sustainable material choices.
- Interdisciplinary Collaboration: Accelerating progress necessitates close collaboration between microbiologists, process engineers, and materials scientists. For example, feedback on the desired material properties and performance from end-users should directly inform the engineering of microbial strains and the design of processing lines [7].
- Focus on Circular Design: Future research must prioritize the development of "drop-in" biopolymers that are compatible with existing recycling infrastructure, as well as designing for biodegradability in specific environments where it offers a clear environmental benefit [47] [48]. This holistic approach ensures that biopolymers fulfill their promise as a cornerstone of a circular economy.
The journey to making biopolymers a mainstream, commercially viable alternative to traditional plastics is complex, yet achievable. By systematically addressing the challenges of scalability and cost through the lens of the materials tetrahedron, researchers and industry professionals can identify targeted solutions. Advancements in processing technologies—such as integrated microfluidic-ultrasonic systems, genetic engineering, and the use of waste feedstocks—are pivotal to tailoring biopolymer structure and enhancing their properties. This, in turn, unlocks superior performance across a wider range of applications. A concerted, interdisciplinary effort that leverages continuous innovation, supportive policies, and circular economy principles is essential to propel biopolymers into a new era of sustainable manufacturing.
Overcoming Information Extraction Hurdles from Scientific Literature
The exponential growth of scientific publications represents a significant bottleneck for researchers, particularly in fields like materials science where global annual publication rates have grown by approximately 59% [54]. This deluge of information makes it increasingly challenging for scientists to stay current with their fields and extract specific insights, such as those related to the materials tetrahedron—the fundamental framework connecting processing, structure, properties, and performance of materials [7]. The Portable Document Format (PDF), accounting for over 83% of documents shared online, has become a particular obstacle to automated knowledge extraction due to its focus on visual presentation rather than machine-readable semantic structure [55]. This technical guide examines current methodologies, challenges, and solutions for extracting structured information from scientific literature, with specific application to materials tetrahedron research, providing researchers with practical frameworks to accelerate discovery.
The Fundamental Challenges in PDF Information Extraction
Extracting structured information from scientific PDFs presents multiple technical hurdles that impact research efficiency in materials science and drug development.
Format and Structural Complexities
PDFs inherently preserve visual layout rather than semantic meaning, creating significant extraction barriers. Documents contain heterogeneous elements including text, tables, figures, and mathematical formulas arranged in complex spatial relationships. This visual fidelity comes at the cost of machine interpretability, as the underlying document structure does not distinguish between these element types or their semantic roles [55]. The conversion process from PDF to analyzable text frequently introduces errors in character recognition, especially with specialized scientific notation and subscripts common in materials science literature. Furthermore, the absence of standardized markup for scholarly concepts means that even successfully extracted text requires substantial post-processing to identify and classify key information entities relevant to the materials tetrahedron framework.
Domain-Specific Adaptation Barriers
Scientific domains employ highly specialized terminologies and conceptual frameworks that challenge general-purpose extraction algorithms. As Kononova et al. (2021) noted, standard natural language processing tools trained on general language data struggle with the specialized vocabulary of scientific publications, particularly in technical fields like materials science [55]. This problem is exacerbated by a critical shortage of annotated domain-specific datasets needed to train and validate extraction models for scientific subfields. The absence of these resources forces research teams to invest substantial time in manual annotation before automated systems can be deployed effectively. Additionally, the rapid evolution of scientific concepts and terminology creates a moving target for extraction systems, requiring continuous adaptation to maintain accuracy.
Table 1: Primary Challenges in Scientific PDF Information Extraction
| Challenge Category | Specific Limitations | Impact on Research Efficiency |
|---|---|---|
| Document Structure | Format preservation over semantic structure; Complex element arrangement; OCR errors with specialized notation | Increases preprocessing overhead; Reduces extraction accuracy for technical content |
| Domain Adaptation | Specialized terminology; Limited annotated datasets; Evolving scientific concepts | Hinders cross-domain application; Requires domain expert involvement; Increases setup time |
| Methodological Limitations | Rule-based system rigidity; Statistical model data demands; LLM hallucinations | Limits adaptability to new document layouts; Constrains application to niche domains; Introduces accuracy concerns |
Current Methodological Approaches
The field of information extraction from scientific literature has evolved through three dominant methodological paradigms, each with distinct strengths and limitations for materials science applications.
Traditional Extraction Methodologies
Early approaches to information extraction relied heavily on manually constructed rules targeting specific document patterns and structures. These rule-based systems utilize predefined patterns, syntactic rules, and document structure heuristics to identify and extract relevant information [55]. While effective for highly standardized document formats with consistent layouts, these systems demonstrate significant rigidity, failing to adapt to variations in document structure or stylistic differences between publications and publishers. Statistical learning-based approaches marked an advancement by applying machine learning models trained on annotated datasets to identify target information. These models typically utilize features such as word frequency, positional information, and lexical patterns to classify text segments [55]. However, they remain constrained by their dependency on substantial volumes of labeled training data, creating a significant bottleneck for application in specialized scientific domains where annotated corpora are scarce.
LLM-Enhanced Extraction Frameworks
The recent advent of large language models has transformed information extraction capabilities, particularly through in-context learning techniques that enable rapid domain adaptation. Modern LLMs can perform sophisticated extractions with minimal task-specific training through two primary approaches: zero-shot learning, where the model performs extraction based solely on task description without examples, and few-shot learning, where the model is provided with a small number of demonstration examples (typically 1-5) illustrating the target extraction [54]. This approach has proven particularly valuable for extracting materials tetrahedron relationships, where models can be directed to identify processing parameters, structural characteristics, material properties, and performance metrics from scientific text. A key advantage of LLM-based extraction is the ability to target diverse semantic concepts within scientific texts—from research questions and methodologies to specific results and conclusions—with minimal domain-specific training [54]. This flexibility enables researchers to rapidly adapt extraction pipelines to target specific aspects of the materials tetrahedron without extensive retraining or system modification.
Table 2: Performance Comparison of Information Extraction Methods
| Extraction Method | Key Advantages | Principal Limitations | Materials Science Applicability |
|---|---|---|---|
| Rule-Based Systems | Predictable results; High precision for targeted patterns; Interpretable logic | Brittle to layout changes; Labor-intensive rule creation; Poor domain transfer | Limited to highly standardized journal formats with consistent terminology |
| Statistical Learning Models | Adaptive to variation; Can learn complex patterns; Reduced manual effort | Requires large annotated datasets; Feature engineering complexity; Domain specificity | Moderate, constrained by limited annotated materials science corpora |
| LLM-Based Approaches | Rapid domain adaptation; Minimal examples required; Broad concept coverage | Computational intensity; Potential hallucinations; Prompt sensitivity | High, particularly for cross-domain extraction of tetrahedron relationships |
Experimental Protocols for Information Extraction
Implementing effective information extraction systems requires methodical experimental design and evaluation frameworks. Below we outline proven protocols for developing and validating extraction pipelines.
Dataset Preparation and Annotation
The foundation of any successful extraction system is a carefully constructed dataset representing the target document types and domains. The PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) methodology provides a rigorous framework for identifying and selecting relevant scientific literature [55]. For materials tetrahedron applications, this begins with formulating targeted search queries combining materials science terminology with tetrahedron concepts (processing-structure-properties-performance). Identified documents must then undergo systematic annotation where domain experts label key entities and relationships. This annotation should specifically target tetrahedron elements: processing parameters (e.g., annealing temperature, synthesis methods), structural characteristics (e.g., microstructure, crystal phase), material properties (e.g., tensile strength, conductivity), and performance metrics (e.g., fatigue resistance, efficiency). Establishing clear annotation guidelines with high inter-annotator agreement is essential for creating reliable gold standard datasets for model training and evaluation.
LLM Implementation and Evaluation
For LLM-based extraction, implementation follows a structured workflow beginning with document preprocessing to convert PDFs to clean text while preserving structural metadata. The extraction pipeline then employs carefully designed prompts that incorporate chain-of-thought reasoning to enhance accuracy [54]. Technical evaluations should compare multiple LLM options, including both commercial and open-source models, using metrics such as precision, recall, and F1-score against manually annotated gold standards [54]. For materials science applications, it is particularly valuable to implement iterative refinement where initial extraction results inform prompt improvements in a cyclic fashion. This approach progressively enhances the system's ability to accurately identify and relate tetrahedron concepts across diverse document types and reporting styles.
A Conceptual Framework for Scientific Information Extraction
To address the limitations of current approaches, we propose a comprehensive framework that integrates multiple components for end-to-end information extraction from scientific PDFs, specifically designed for materials tetrahedron research.
Framework Architecture and Components
This conceptual framework comprises nine integrated modules that work in concert to transform unstructured PDF content into structured knowledge. The document manager handles ingestion and storage of scientific papers, while the pre-processor performs critical cleanup tasks including text extraction, OCR for scanned documents, and section identification. An ontology manager provides domain-specific knowledge structures, including formal representations of the materials tetrahedron framework, to guide the extraction process [55]. The core information extractor module employs adaptable techniques (LLM-based, rule-based, or hybrid) to identify target entities and relationships. Additional components include an annotation engine to support manual validation and correction, a question-answering tool for interactive querying of extracted content, a knowledge visualizer to represent extracted relationships graphically, and a data exporter to format results for various downstream applications. This modular architecture ensures the system can evolve with changing extraction requirements and adapt to new scientific subdomains.
Implementation Considerations for Materials Science
Successful implementation for materials science applications requires specific adaptations to address domain-specific challenges. The framework must incorporate specialized ontologies covering materials science concepts, processing techniques, characterization methods, and property classifications. The extraction targets should explicitly focus on identifying and linking tetrahedron elements—for example, connecting specific thermal processing parameters (processing) to resulting microstructural features (structure), then to measured mechanical properties (properties), and ultimately to performance under specific conditions (performance). The system should implement human-in-the-loop validation where materials science experts review and correct critical extractions, with these corrections feeding back to improve the extraction models. Additionally, integration with existing materials knowledge graphs, such as those developed by the Open Research Knowledge Graph initiative, can enhance connectivity and discovery across the extracted information [54].
Table 3: Research Reagent Solutions for Information Extraction
| Tool Category | Specific Solutions | Function in Extraction Pipeline |
|---|---|---|
| LLM Platforms | Gemini 1.5 Flash & Pro; GPT-4; Llama 3.3 70B; Qwen 2.5 72B | Semantic analysis and concept extraction via in-context learning [54] |
| Document Processing | Custom PDF parsers; OCR engines (Tesseract); Layout analysis tools | Text extraction from diverse PDF formats; Handling scanned documents [55] |
| Knowledge Representation | Open Research Knowledge Graph; Domain-specific ontologies | Structuring extracted information; Enabling semantic search and integration [54] |
| Evaluation Frameworks | Precision/recall metrics; Domain expert validation; Task-specific benchmarks | Performance measurement; Quality assurance; System improvement guidance [55] |
Application to Materials Tetrahedron Research
The materials tetrahedron framework provides a powerful conceptual structure for organizing extraction efforts in materials science, with specific applications accelerating research and development.
Extracting Tetrahedron Relationships
In materials tetrahedron research, information extraction systems can be targeted to identify and connect critical relationships between processing parameters, resulting structures, material properties, and ultimate performance characteristics. For polyhydroxyalkanoate biopolymers, for example, extraction systems can identify how synthesis conditions (processing) influence crystalline morphology (structure), which determines biodegradation rates (properties) relevant to specific applications (performance) [7]. This approach enables the systematic population of structure-property-processing-performance databases from existing literature, creating valuable resources for materials design and selection. Advanced extraction systems can even identify implicit relationships within texts where authors describe but do not explicitly connect tetrahedron elements, using linguistic patterns and contextual analysis to reconstruct complete relationships from fragmented information across multiple publications.
Accelerating Materials Discovery
The TETRA (Transforming Evaluation and Testing via Robotics and Acceleration) initiative at Johns Hopkins Applied Physics Laboratory demonstrates how extracted knowledge can accelerate materials development. This approach integrates robotics, artificial intelligence, and accelerated synthesis to rapidly explore material compositions and processing parameters [3]. By extracting existing knowledge from literature, researchers can inform AI models that recommend promising experimental directions, effectively creating AI "co-investigators" that learn from materials development data to guide research [3]. This integration of extracted knowledge with experimental automation represents a powerful paradigm for reducing materials development timelines from years to months or weeks, particularly for mission-critical applications in defense and healthcare.
Emerging Trends and Technologies
The field of scientific information extraction is rapidly evolving, with several emerging trends poised to address current limitations. Multi-modal extraction approaches that combine textual analysis with figure and table interpretation will enable more comprehensive knowledge capture from scientific documents. For materials science specifically, this includes extracting data from microscopy images, diffraction patterns, and property graphs. Autonomous experimental design represents another frontier, where extraction systems identify knowledge gaps in the literature and actually propose hypothesis-driven experiments to address them [3]. The development of cross-domain foundation models specifically pretrained on scientific literature will reduce the dependency on domain-specific fine-tuning, while federated learning approaches may help overcome data scarcity issues by allowing models to learn from multiple institutions without sharing proprietary data. As these technologies mature, we anticipate increasingly sophisticated systems capable of not just extracting known relationships but discovering novel connections and hypotheses across the materials tetrahedron.
Information extraction from scientific literature has evolved from rigid rule-based systems to adaptable LLM-powered frameworks capable of capturing the complex relationships encapsulated in the materials tetrahedron. While significant challenges remain in document structure interpretation, domain adaptation, and evaluation, current methodologies already offer substantial value for accelerating materials research and drug development. The integration of robust extraction pipelines with knowledge graphs and experimental design systems creates a powerful infrastructure for scientific discovery. As these technologies continue to mature, they will increasingly serve as force multipliers for researchers, enabling more efficient navigation of the scientific literature and accelerating the translation of published knowledge into practical innovations. For materials scientists and research professionals, adopting these extraction methodologies represents a strategic imperative for maintaining competitiveness in an era of exponentially growing publication volumes.
The integration of Artificial Intelligence (AI) and robotics is initiating a paradigm shift in materials science and drug development. This transformation is strategically addressing some of the most persistent challenges in these fields: lengthy development timelines, high costs, and low success rates. By creating closed-loop, automated systems, researchers can now navigate the complex relationships of the materials science tetrahedron (MST)—which links a material's processing, structure, properties, and performance—with unprecedented speed and precision [3] [56] [2]. This whitepaper delves into the core methodologies of this new paradigm, exemplified by pioneering programs like the TETRA initiative, and provides a detailed technical guide for researchers and scientists aiming to implement these accelerated development techniques.
The Foundation: Materials Science Tetrahedron in the Age of Acceleration
The materials science tetrahedron (MST) provides a fundamental conceptual framework for understanding the interdependent relationship between the processing, structure, properties, and performance of any material [2]. In pharmaceuticals, this translates to the development of drug products where the crystal form, formulation, and manufacturing process directly dictate the therapeutic efficacy and safety of the final product [2].
Traditionally, exploring these relationships has been a slow, sequential, and resource-intensive process. Scientists were forced to investigate one variable at a time, with cycles often taking months and requiring the production of large material ingots [3]. The new paradigm, championed by efforts like the Johns Hopkins Applied Physics Laboratory (APL) TETRA program, reimagines this approach. It leverages AI and robotics to simultaneously explore the vast parameter space of the tetrahedron [3]. This is achieved by integrating advanced computational models with high-throughput, robotic experimentation, effectively creating a continuous R&D loop that dramatically accelerates the journey from material concept to optimized component or drug formulation [3] [57].
Core AI and Robotic Technologies
Artificial Intelligence and Machine Learning
AI and Machine Learning (ML) serve as the central nervous system for accelerated development, enabling data-driven decision-making and predictive modeling.
- Physics-Informed Machine Learning: These models incorporate fundamental physical laws and constraints into the learning process, ensuring predictions are not only data-driven but also scientifically plausible. For instance, the Max Planck Institute employs interpretable, physics-informed ML models to predict and optimize the strength of complex high-entropy alloys, bringing much-needed transparency to AI recommendations [58].
- Active Learning and Closed-Loop Discovery: This AI strategy involves a continuous cycle of prediction, experimentation, and learning. The ML model identifies the most promising candidates or experiments from a vast pool of possibilities, which are then synthesized and tested. The results from these tests are fed back into the model to refine its future predictions. This approach has been successfully used to identify high-entropy Invar alloys with exceptionally low thermal expansion coefficients [58].
- Large Language Models (LLMs) for Knowledge Mining: LLMs are being deployed to extract critical insights and design principles from the vast, unstructured data of scientific literature. One project analyzed over 6 million research articles to identify previously undiscovered compositions for high-performance alloys, a task infeasible for human researchers alone [58].
- AI for Advanced Data Analysis: AI frameworks, particularly deep learning models like 3D convolutional neural networks (CNNs), are revolutionizing the analysis of complex experimental data from techniques such as Atom Probe Tomography (APT) and X-ray Diffraction (XRD), achieving high accuracy even with noisy data [58].
Robotic and Automation Systems
Robotic systems act as the hands of the operation, executing high-throughput experiments with superhuman precision and endurance.
- Combinatorial Synthesis via Additive Manufacturing: The TETRA program utilizes techniques like blown-powder directed energy deposition (DED) to fabricate hundreds of distinct alloy variants on a single build plate. This allows for the rapid exploration of chemical compositions and the creation of custom-designed 3D specimens ready for autonomous testing [3].
- Automated Laboratory Robotics: In pharmaceutical research, robotic systems automate repetitive but critical tasks such as high-throughput screening (HTS), sample preparation, liquid handling, and testing [59] [60]. These systems can operate 24/7, drastically increasing experimental throughput.
- Collaborative Robots (Cobots): Unlike traditional industrial robots, cobots are designed to work safely alongside human researchers. They are particularly valuable in laboratories for tasks requiring flexibility and precision, such as the small-batch production of personalized medicines [60] [61].
- Self-Running Laboratories: The ultimate expression of this integration is the "self-running lab," where an AI "co-engineer" manages the robotic systems to autonomously design experiments, synthesize materials, test them, and analyze the results, continuously iterating towards a defined goal [3].
Detailed Experimental Protocols
Protocol 1: High-Throughput Alloy Discovery and Optimization (TETRA Framework)
This protocol outlines the steps for accelerated discovery of metallic components, as exemplified by the TETRA program [3].
1. Objective: To rapidly discover and optimize a new alloy with target mechanical properties (e.g., high tensile strength, corrosion resistance) for a specific mission-critical application.
2. Experimental Workflow:
- Step 1: AI-Driven Design Space Exploration.
- Methodology: Use an AI platform (e.g., ExoMatter, Materials Project) to screen existing databases of inorganic materials based on target performance, sustainability, and cost [62]. Train a physics-informed ML model on existing data to predict the properties of unseen compositions and suggest initial candidate compositions [58].
- Step 2: Robotic Combinatorial Synthesis.
- Methodology: Employ a blown-powder Directed Energy Deposition (DED) additive manufacturing system. The system is programmed to fabricate an array of hundreds of discrete test specimens on a single build plate, with each specimen having a systematically varied chemical composition and/or microstructure based on the AI's suggestions [3].
- Step 3: Automated Post-Processing.
- Methodology: Transfer the entire build plate to integrated, robotic post-processing stations. This may include custom heat treatment furnaces and hot forging equipment to modify the microstructure and properties of the printed specimens, accounting for critical production process effects [3].
- Step 4: Robotic Mechanical Property Measurement.
- Methodology: A robotic arm autonomously transfers individual test specimens from the build plate to a mechanical testing system (e.g., tensile tester, hardness indenter). The system conducts the tests and records the property data (e.g., yield strength, elongation) for each specimen without human intervention [3].
- Step 5: Data Integration and Active Learning.
- Methodology: The property data from testing is automatically fed back into the AI model. The model uses an active learning strategy to analyze the new data, update its predictions, and recommend the next, more optimal set of compositions and processing parameters to be synthesized and tested, closing the loop [3] [58].
High-Throughput Alloy Discovery Workflow
Protocol 2: AI-Guided Pharmaceutical Formulation Development
This protocol describes the use of AI and robotics to accelerate the development of optimal drug formulations, addressing low clinical success rates [57].
1. Objective: To identify a stable and bioavailable formulation for a New Chemical Entity (NCE) with poor aqueous solubility.
2. Experimental Workflow:
- Step 1: Preformulation Data Mining and Virtual Screening.
- Methodology: Use AI tools to mine scientific literature and internal databases for successful formulation strategies for chemically similar compounds. Deploy ML models to virtually screen and predict the compatibility of various API crystal forms (polymorphs, salts, co-crystals) and excipients [58] [57].
- Step 2: High-Throughput Formulation Preparation.
- Methodology: Utilize robotic liquid handlers and automated systems (e.g., ESSERT MicroFactory) to prepare hundreds of micro-batch formulations. These systems precisely dispense API and excipients in varying ratios to create a wide array of formulations, such as solid dispersions or lipid-based systems [59] [61].
- Step 3: Automated In-Vitro Characterization.
- Methodology: Robotic arms transfer the prepared formulations to integrated analytical instruments for high-throughput characterization. Key tests include:
- Dissolution Testing: Automated dissolution apparatuses with in-line UV or HPLC analysis to profile drug release.
- Stability Assessment: Robotic placement of samples into stability chambers under controlled conditions (e.g., temperature, humidity).
- Solid-State Analysis: Automated XRPD or Raman spectroscopy to confirm crystal form and detect amorphous content [57] [61].
- Methodology: Robotic arms transfer the prepared formulations to integrated analytical instruments for high-throughput characterization. Key tests include:
- Step 4: Data Analysis and Optimization Loop.
- Methodology: ML algorithms correlate the formulation composition and processing parameters (MST's "Processing") with the characterized properties (MST's "Properties") and predicted performance (MST's "Performance"). The model then identifies the formulation that best meets the target product profile and may recommend a focused set of confirmatory experiments [57].
AI-Guided Pharmaceutical Formulation Workflow
Quantitative Data and Performance Metrics
The integration of AI and robotics delivers measurable, transformative improvements in R&D efficiency and output quality. The tables below summarize key quantitative gains.
Table 1: Performance Metrics of AI and Robotic Integration in R&D
| Metric | Traditional Approach | AI/Robotics Approach | Improvement | Source |
|---|---|---|---|---|
| Materials Development Cycle Time | Months per iteration | Days per iteration | Reduction from months to days | [3] |
| Pharmaceutical R&D Time & Cost | Baseline | AI-driven discovery to preclinical | 25 - 50% savings | [57] |
| Production Throughput | Manual process baseline | 24/7 robotic operation | 30 - 50% increase | [61] |
| Product Defect Rate | Baseline manual error rate | Robotic precision | Up to 80% reduction | [61] |
| Workplace Accident Rate | Baseline manual handling | Automation of hazardous tasks | Up to 70% reduction | [61] |
Table 2: Capabilities of AI Models in Materials Characterization
| AI Model / Technique | Application | Key Performance Metric | Source |
|---|---|---|---|
| Residual Hybrid Learning Model (RELM) | Predicting strength of high-entropy alloys | High predictive accuracy with sparse, skewed datasets; provides interpretable insights. | [58] |
| 3D Convolutional Neural Network (CNN) | Crystal structure recognition from Atom Probe Tomography data | >98% accuracy, even with random displacements and missing atoms. | [58] |
| Active Learning with DFT/Thermodynamics | Discovering high-entropy Invar alloys | Identified 2 optimal alloys from millions of candidates via closed-loop feedback. | [58] |
| Large Language Models (LLMs) | Mining research articles for new compositions | Analyzed 6+ million articles to identify previously undiscovered alloy systems. | [58] |
The Scientist's Toolkit: Key Research Reagent Solutions
Implementing the advanced protocols described requires a suite of essential computational and physical tools. The following table details these key resources.
Table 3: Essential Resources for AI and Robotics-Accelerated R&D
| Tool / Resource | Function | Relevance to MST | |
|---|---|---|---|
| The Materials Project Database | A free database of computed material properties (e.g., elasticity, band structure) for over 160,000 inorganic compounds. Provides data for training AI/ML models. | Links atomic-level Structure to predicted Properties, guiding the design of new materials. | [56] |
| Directed Energy Deposition (DED) Additive Manufacturing | A robotic blown-powder 3D printing system that enables combinatorial synthesis of metal samples with graded compositions and microstructures. | The primary tool for implementing Processing, creating varied Structures for study. | [3] |
| Physics-Informed Machine Learning Models | ML algorithms that incorporate physical laws as constraints, making predictions more interpretable and physically plausible. | Maps the relationship between Processing parameters, material Structure, and resulting Properties. | [58] |
| Robotic Mechanical Testing System | An automated system that physically transfers test specimens to a load frame, conducts tests (e.g., tensile, compression), and records data without human intervention. | Directly measures the Properties (e.g., strength) that determine real-world Performance. | [3] |
| High-Throughput Screening (HTS) Robotic Systems | Automated workstations for liquid handling, assay preparation, and sample management, widely used in pharmaceutical labs. | Rapidly tests how formulation Processing (composition) affects drug release Properties and Performance. | [59] [60] |
The strategic convergence of AI, robotics, and the foundational principles of the materials science tetrahedron marks the beginning of a new era in materials science and pharmaceutical development. This paradigm, as demonstrated by the TETRA program and other leading research, moves beyond slow, sequential experimentation to a dynamic, data-driven, and highly parallelized approach. By adopting these advanced techniques—closing the loop between AI-driven design and robotic experimentation—researchers and drug development professionals can dramatically accelerate the discovery and optimization of critical materials and life-saving therapeutics. This not only promises enhanced efficiency and cost savings but also a higher probability of clinical success, ultimately delivering innovative solutions to market faster.
The discovery and development of advanced materials are fundamental to technological progress across industries, from aerospace to drug development. The materials science tetrahedron provides an essential paradigm for understanding the complex, interdependent relationships between a material's processing, its resulting structure across multiple length scales, its fundamental properties, and its ultimate performance in application [63]. This framework establishes that performance is not an isolated outcome but emerges from the careful balancing of these four interconnected elements.
Optimizing for multiple constraints—specifically balancing desired properties with manufacturability and degradation characteristics—represents one of the most significant challenges in modern materials design. Conventional approaches often prioritize immediate performance metrics at the expense of other critical factors, leading to materials that may excel in limited laboratory conditions but fail in real-world applications due to poor manufacturability, unpredictable degradation, or environmental incompatibility. This guide examines systematic approaches for navigating these complex trade-offs, with particular emphasis on methodologies applicable to biomedical and sustainable material development. By adopting an integrated perspective grounded in the materials tetrahedron, researchers can develop more sophisticated strategies for creating materials that successfully balance multiple, often competing, requirements.
The Materials Tetrahedron: A Framework for Multi-Constraint Optimization
Core Components and Interrelationships
The materials tetrahedron defines the scope of materials science and engineering through four interdependent aspects: processing, structure, properties, and performance [63]. These elements exist in a continuous cycle of influence, where each corner of the tetrahedron affects and is affected by the others. Processing encompasses all methods used to synthesize and shape a material, from initial synthesis to final manufacturing steps. Structure refers to the material's arrangement at all length scales, from atomic bonding to microscopic features and macroscopic architecture. Properties are the material's responses to external stimuli, including mechanical, thermal, electrical, and chemical characteristics. Performance represents how effectively the material functions in its intended application under real-world conditions [63].
The tetrahedron framework is particularly valuable for visualizing and managing trade-offs in materials design. For example, a processing change to improve manufacturability may alter the material's structure in ways that affect both its properties and degradation behavior. Similarly, optimizing for a specific performance metric often requires compromising on other properties or accepting more complex manufacturing requirements. The tetrahedron provides a mental model for tracing these interconnected effects throughout the materials development cycle.
Visualizing the Tetrahedron Framework
The following diagram illustrates the core relationships of the materials science tetrahedron, including the central role of characterization in connecting these elements:
Key Challenges in Balancing Multiple Constraints
Property-Processing Trade-offs in Material Design
Achieving optimal material properties often requires processing conditions that conflict with manufacturability constraints. For example, certain thermal treatments may enhance mechanical properties but introduce dimensional instability or residual stresses that complicate manufacturing. In metallic systems, rapid cooling can produce desirable fine-grained microstructures but may also cause distortion or cracking that limits design freedom. Similar challenges appear in polymer systems, where processing conditions for achieving specific molecular orientations often conflict with requirements for uniform dimensional control [13] [63].
The property-processing relationship is particularly pronounced in multi-material additive manufacturing (MMAM), where different materials within a single component may require incompatible processing parameters. Research has demonstrated that material interface challenges represent a significant barrier to implementing MMAM, as differential thermal expansion, residual stresses, and weak interfacial bonding can compromise mechanical performance [64] [65]. These challenges necessitate sophisticated approaches to manage the trade-offs between achieving desired properties and maintaining manufacturability.
Degradation-Performance Balancing in Specific Applications
Materials designed for biomedical applications or environmental sustainability must balance functional performance with controlled degradation profiles. This challenge is exemplified in the development of polyhydroxyalkanoates (PHAs), a family of biologically produced polyesters investigated as alternatives to conventional plastics [13]. While PHAs offer attractive biodegradability and biocompatibility, their limited chemical diversity results in a narrow range of thermal processing windows and mechanical properties that can restrict their application. The degradation rate of PHAs must be carefully balanced against their functional lifetime requirements—too rapid degradation compromises performance, while too slow degradation limits their value as sustainable alternatives [13].
Similar challenges appear in pharmaceutical development, where drug delivery systems must maintain structural integrity until reaching target sites while degrading predictably to release therapeutic agents. This requires precise control over degradation mechanisms in response to specific environmental triggers, balanced against the mechanical properties needed for manufacturing and deployment.
Economic and Scalability Constraints
Beyond technical considerations, economic factors frequently constrain materials optimization. The high cost of commercially available PHAs (approximately $1.81–3.20 per lb compared to $0.45–0.68 per lb for polypropylene) illustrates how processing challenges and limited production scale can restrict practical application, even for materials with theoretically attractive property profiles [13]. Similar economic constraints appear in pharmaceutical development, where manufacturing complexity can determine whether a promising material progresses from laboratory research to clinical application.
Scaling laboratory-proven MMAM techniques to industrial applications presents significant challenges, including material interface issues, environmental durability concerns, and the absence of design tools specific to building-scale components [65]. These challenges highlight the importance of considering scalability throughout the optimization process rather than as an afterthought.
Case Study: Optimizing Polyhydroxyalkanoate (PHA) Biopolymers
PHA Processing-Structure-Property Relationships
Polyhydroxyalkanoates represent an instructive case study in multi-constraint optimization within the materials tetrahedron framework. As biologically produced polyesters, PHAs are synthesized by microorganisms in the presence of excess carbon sources, acting as an energy storage mechanism [13]. This biological origin fundamentally shapes their processing-structure-property relationships, presenting both advantages and constraints for practical application.
The processing of PHAs begins with biosynthesis, where factors including microorganism selection, nutrient availability, and cultivation conditions determine the polymer composition and molecular weight. Post-synthesis processing such as melt extrusion, compression molding, or solvent casting further influences the material's structure by affecting crystallinity, orientation, and morphology [13]. These structural features directly determine key properties including thermal stability, mechanical strength, and degradation rate. The following table summarizes key PHA types and their properties:
Table 1: Commercial Polyhydroxyalkanoates (PHAs) and Their Properties
| PHA Type | Chemical Structure | Key Properties | Processing Challenges | Degradation Characteristics |
|---|---|---|---|---|
| PHB (P3HB) | Homopolymer of 3-hydroxybutyrate | High crystallinity, brittleness, high melting point (~175°C) | Narrow processing window, thermal degradation during melting | Slow degradation in ambient conditions; faster in compost |
| PHBV (P3HB3HV) | Copolymer of 3-hydroxybutyrate and 3-hydroxyvalerate | Reduced crystallinity, improved toughness, lower melting point | Broader processing window than PHB | Tunable degradation rate based on valerate content |
| PHBHHx | Copolymer of 3-hydroxybutyrate and 3-hydroxyhexanoate | Increased flexibility, reduced crystallinity | Improved processability | Enhanced biodegradability in various environments |
Experimental Protocol: Processing PHAs for Controlled Degradation
Objective: To process PHA biopolymers with controlled degradation profiles while maintaining mechanical integrity for target applications.
Materials and Equipment:
- PHA polymer (e.g., PHB, PHBV, or PHBHHx)
- Plasticizers (e.g., citrate esters, polyethylene glycol)
- Compatibilizers for blend systems
- Twin-screw extruder with temperature control
- Injection molding machine or compression press
- Differential scanning calorimetry (DSC) instrument
- Thermogravimetric analyzer (TGA)
- Universal testing machine
- Controlled degradation environment (compost, simulated marine, or enzymatic solution)
Methodology:
Material Preparation:
- Pre-dry PHA pellets at 60°C for 24 hours to remove moisture
- For blend systems, pre-mix PHA with additives using a high-speed mixer
Melt Processing:
- Process using twin-screw extruder with temperature profile optimized for specific PHA type
- For PHB: 160-175°C (feed to die zones)
- For PHBV: 150-170°C (feed to die zones)
- For PHBHHx: 140-160°C (feed to die zones)
- Maintain screw speed at 50-100 rpm to control shear history
- Collect extrudate, water-cool, and pelletize
Forming:
- Injection mold test specimens using temperature profile 10-15°C below extrusion maximum
- Apply moderate injection speed and hold pressure to minimize orientation
Characterization:
- Determine thermal properties using DSC (heating rate 10°C/min under nitrogen)
- Assess thermal stability using TGA (heating rate 20°C/min under nitrogen)
- Evaluate mechanical properties according to ASTM standards
- Perform degradation studies in controlled environments with periodic assessment of weight loss and property changes
Key Processing Parameters:
- Maximum processing temperature and thermal residence time
- Cooling rate after processing
- Presence of nucleating agents or plasticizers
- Molecular weight and purity of the PHA
This protocol enables systematic investigation of processing-structure-property-degradation relationships in PHA biopolymers, facilitating optimization for specific application requirements.
Optimization Workflow for PHA Development
The following diagram illustrates the iterative optimization workflow for developing PHA materials with balanced properties, processability, and degradation:
Advanced Approaches for Multi-Constraint Optimization
Multi-Material Additive Manufacturing (MMAM) Strategies
Multi-material additive manufacturing represents a transformative approach for balancing multiple constraints by enabling the spatial distribution of different materials within a single component [64] [65]. This capability allows designers to locate specific properties where they are most needed, optimizing overall performance while addressing manufacturability and degradation requirements.
In MMAM, functionally graded materials (FGMs) with continuous property transitions can be created to manage stress concentrations, thermal gradients, or degradation fronts [65]. For example, a biomedical implant might transition from a stiff material at load-bearing surfaces to a porous, biodegradable material that promotes tissue integration. Similarly, architectural components might integrate structural and insulating materials within a single manufacturing process, optimizing both mechanical and thermal performance [65].
Key challenges in MMAM include managing material interfaces, ensuring compatibility between different materials, and developing design tools that can effectively model the behavior of multi-material systems. Research priorities identified for advancing MMAM include developing integrated optimization frameworks, multiscale modeling techniques, novel material combinations, and standardized protocols for evaluating long-term performance [65].
Data-Driven and AI-Enabled Optimization
Machine learning and artificial intelligence are increasingly important for navigating complex design spaces with multiple constraints. These approaches can identify non-intuitive relationships between processing parameters, material structure, and resulting properties that might be overlooked using traditional experimental methods.
The Tabular Prior-data Fitted Network (TabPFN) represents a significant advancement in data-driven materials optimization [66]. This tabular foundation model outperforms traditional methods like gradient-boosted decision trees on datasets with up to 10,000 samples, using substantially less training time. TabPFN uses in-context learning—the same mechanism that enables large language models to perform complex reasoning tasks—to generate powerful tabular prediction algorithms that are fully learned [66].
For materials researchers, such AI tools can accelerate the discovery of processing parameters that balance multiple constraints, predict degradation behavior based on structural features, or identify material compositions with optimized property profiles. The following table summarizes computational approaches for multi-constraint optimization:
Table 2: Computational Approaches for Multi-Constraint Materials Optimization
| Method | Key Features | Applications | Limitations |
|---|---|---|---|
| TabPFN | Transformer-based foundation model for tabular data; fast inference on small-to-medium datasets | Property prediction, processing parameter optimization, degradation modeling | Limited to datasets with <10,000 samples; requires relevant feature set |
| Functionally Graded Material Optimization | AI-driven optimization of material distribution within a component | Stress reduction, thermal management, controlled degradation | Computationally intensive for complex geometries |
| Multi-scale Modeling | Links models across different length scales (atomic to macroscopic) | Predicting processing-microstructure relationships, property prediction | Requires significant computational resources; validation challenges |
| Bayesian Optimization | Efficient exploration of high-dimensional parameter spaces | Experimental design, process optimization | Performance depends on choice of surrogate model and acquisition function |
Integrated Workflow for AI-Assisted Materials Optimization
The following diagram illustrates how AI and machine learning can be integrated into the materials development workflow to balance multiple constraints:
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Reagents for Multi-Constraint Materials Research
| Material/Reagent | Function in Research | Application Examples | Considerations for Multi-Constraint Optimization |
|---|---|---|---|
| PHA Biopolymers (PHB, PHBV, PHBHHx) | Model biodegradable polymer system for studying structure-property-degradation relationships | Sustainable packaging, biomedical implants, drug delivery systems | Balance mechanical properties with degradation rate; optimize processing window |
| Compatibilizers (e.g., maleic anhydride grafted polymers) | Improve interfacial adhesion in polymer blends and composites | Multi-material systems, composite materials | Impact on both processing behavior and final properties; potential effect on degradation |
| Plasticizers (citrate esters, PEG) | Modify processing characteristics and mechanical properties | Improving processability of brittle biopolymers | Potential migration issues; effect on degradation kinetics |
| Nucleating Agents (boron nitride, talc) | Control crystallization behavior during processing | Managing crystallinity in semi-crystalline polymers | Influence on mechanical properties and degradation behavior |
| Crosslinking Agents (peroxides, silanes) | Modify network structure to control properties | Enhancing thermal stability or mechanical properties | Effect on processability and degradation profile |
| Bioactive Fillers (hydroxyapatite, bioactive glass) | Add functionality to base materials | Bone tissue engineering, controlled release systems | Impact on processing parameters and interface stability during degradation |
Optimizing materials for multiple constraints requires a holistic approach that considers the complete materials tetrahedron—recognizing that processing affects structure, structure determines properties, and properties ultimately control performance. This integrated perspective enables researchers to make informed trade-offs rather than treating constraints as isolated problems. The case of PHA biopolymers demonstrates how systematic investigation of processing-structure-property relationships can lead to materials with improved balance between mechanical performance, manufacturability, and degradation characteristics.
Future advances in multi-constraint optimization will likely come from several complementary directions. Multi-material additive manufacturing will enable more sophisticated spatial control of properties within components, allowing designers to locate specific characteristics where they are most needed. AI-driven methods like TabPFN will accelerate the discovery of processing parameters and material compositions that balance competing requirements. Additionally, improved characterization techniques and modeling approaches will provide deeper insights into the fundamental relationships between material structure, properties, and degradation behavior.
For researchers and drug development professionals, adopting these integrated optimization strategies will be essential for developing next-generation materials that meet increasingly complex performance requirements while addressing manufacturability, economic, and sustainability constraints. By framing materials development within the tetrahedron paradigm and leveraging emerging computational and experimental tools, the materials science community can continue to advance the design of sophisticated materials systems optimized for multiple constraints.
Proving the Paradigm: Validation, Comparative Analysis, and Readiness Assessment
In the rigorous field of research and development, particularly within materials science and pharmaceutical development, structured validation frameworks are indispensable for objectively assessing progress and mitigating risk. Two pivotal concepts in this domain are Technology Readiness Levels (TRLs) and Material Maturation Levels (MMLs). TRLs provide a common set of definitions for determining the maturity of a technology, from basic research to proven application [67]. When applied specifically to the materials tetrahedron—a conceptual framework illustrating the dynamic interrelationships between a material's processing, structure, and properties, which collectively determine its performance—the need for a complementary Material Maturation Level (MML) framework becomes evident. While TRLs are well-established for technologies like medical countermeasures [68] and devices [69], MMLs represent a more specialized focus on the maturation journey of a material itself. These frameworks offer researchers, scientists, and drug development professionals a standardized lexicon and evidence-based milestones to track development, communicate status to stakeholders, and make critical go/no-go decisions regarding resource allocation [70]. In the context of the materials tetrahedron, they ensure that advancements in processing, revelations about structure, and enhancements in properties are systematically validated before being relied upon for critical performance outcomes.
Technology Readiness Levels (TRLs): A Detailed Analysis
Core Principles and Definitions
The Technology Readiness Level (TRL) scale is a systematic metric, originally developed by NASA, used to assess the maturity of a particular technology. It consists of nine discrete levels, each representing a specific stage of development, from basic principles (TRL 1) to full deployment (TRL 9) [70]. The fundamental premise of the TRL scale is its evidence-based approach; progression to a higher TRL requires demonstrated validation through prototype testing, documentation, or proven performance in a relevant environment [70]. This framework provides a common language for engineers, scientists, and project managers, enabling clear communication about a technology's status across different disciplines and organizations. For high-stakes industries like pharmaceuticals and defense, this clarity is crucial for managing risk, as the GAO has stated that the maturity of a new technology at the time of its incorporation into a product is directly correlated with the program's ultimate success [69].
TRLs in Pharmaceutical and Medical Development
In the biomedical and pharmaceutical sectors, the generic TRL scale has been adeptly tailored to align with rigorous regulatory pathways, such as those governed by the U.S. Food and Drug Administration (FDA). The development of medical countermeasures (drugs and biologics) follows a well-defined TRL trajectory, where each level is anchored to specific, completed research activities [68].
The table below summarizes the TRLs for medical countermeasure development, integrating key activities from discovery to post-approval [68].
Table 1: Technology Readiness Levels for Medical Countermeasure Development
| TRL | Stage Name | Key Activities and Milestones |
|---|---|---|
| 1 | Review of Scientific Knowledge Base | Active monitoring of scientific literature; assessment of foundational knowledge. |
| 2 | Development of Hypotheses & Designs | Generation of research ideas and experimental designs via "paper studies." |
| 3 | Target/Candidate Identification & Characterization | Basic research begins; in vitro activity demonstrated; preliminary in vivo proof-of-concept (non-GLP). |
| 4 | Candidate Optimization & Non-GLP In Vivo Demo | Non-GLP in vivo efficacy and toxicity studies; initiate animal model and assay development; manufacture lab-scale product. |
| 5 | Advanced Characterization & GMP Process Initiation | Continue non-GLP studies; establish draft Target Product Profile (TPP); initiate scalable GMP process development. |
| 6 | GMP Pilot Lot, IND Submission, & Phase 1 Trial | Manufacture GMP pilot lot; submit Investigational New Drug (IND) application; complete Phase 1 clinical trial for safety. |
| 7 | Scale-up, GMP Validation, & Phase 2 Trial | Scale-up and validate GMP process; conduct GLP animal efficacy studies; complete Phase 2 clinical trial. |
| 8 | GMP Validation Completion, Pivotal Trials, & FDA Approval | Complete pivotal (e.g., Phase 3) trials; submit and obtain FDA approval via NDA/BLA. |
| 9 | Post-Approval Activities | Conduct post-marketing (Phase 4) studies and safety surveillance. |
Similarly, the medical device industry employs a TRL framework that integrates with FDA regulatory milestones like the Investigational Device Exemption (IDE) and Premarket Approval (PMA). For instance, reaching TRL 6 for a Class III device is defined by data from an initial clinical investigation demonstrating safety and supporting progression to larger trials [69]. This alignment with regulatory checkpoints provides a pragmatic roadmap for developers, emphasizing that risk reduction is not linear and often only becomes significant after the completion of clinical trials [69].
Material Maturation Levels (MMLs) and the Materials Tetrahedron
The Materials Tetrahedron as a Foundational Framework
The materials tetrahedron is a central paradigm in materials science, representing the fundamental, interdependent relationship between four key elements: Processing, Structure, Properties, and Performance. This framework posits that a material's performance in a real-world application is not an isolated outcome but the direct result of its properties. These properties are governed by the material's internal structure across multiple length scales (atomic, micro-, macro-), which is, in turn, dictated by the processing methods used to create and shape the material [3]. This is not a linear sequence but a dynamic feedback loop; understanding performance can lead to revised property requirements, which necessitate the development of new processing techniques to achieve novel structures. The TETRA initiative at Johns Hopkins APL, which aims to accelerate materials development, is explicitly founded on reimagining this traditional tetrahedron by integrating robotics and artificial intelligence to explore all these variables simultaneously rather than serially [3].
Conceptualizing Material Maturation Levels (MMLs)
While the search results do not provide a standardized definition for "Material Maturation Levels" (MMLs) analogous to TRLs, the concept can be logically derived from the principles of the materials tetrahedron and the observed need for specialized readiness frameworks. An MML framework would be designed to quantitatively assess the maturity of a material itself, tracking its journey from a novel composition to a reliably characterized and qualified component ready for a specific application.
The core differentiator between TRLs and MMLs lies in their focus. A TRL assesses the maturity of an integrated technology or product (e.g., a drug, a device), whereas an MML would assess the maturity of the material that constitutes a critical component of that technology. For example, a novel biodegradable polymer for controlled drug release would have its own MML, while the final drug-product device would be tracked via TRL.
Table 2: Proposed Framework for Material Maturation Levels (MMLs)
| Proposed MML | Focus Area | Evidence of Maturation |
|---|---|---|
| 1 | Fundamental Concept | Theoretical prediction of a material composition or structure with potential to yield desired properties. |
| 2 | Initial Synthesis & Isolation | Laboratory-scale synthesis of the material with basic characterization confirming primary chemical structure. |
| 3 | Property Validation | Experimental data demonstrating key properties relevant to the target performance in a controlled environment. |
| 4 | Processing-Property Relationships | Understanding of how key processing parameters (e.g., heat treatment, synthesis path) influence critical properties. |
| 5 | Scalable Production & Consistency | Development of a scalable, reproducible synthesis/production process with demonstrated batch-to-batch consistency. |
| 6 | Performance & Durability Testing | Validation of performance and long-term stability (durability) under simulated operational conditions. |
| 7 | Prototype Integration & Field Testing | Successful integration into a subsystem or prototype and validation of performance in a relevant operational environment. |
| 8 | Qualification & Certification | Completion of all required qualification testing and receipt of necessary material certifications for the application. |
| 9 | Field-Proven Performance | Extensive, successful deployment in the final application with a track record of reliable performance. |
Synergistic Application of TRLs and MMLs in Research
An Integrated Workflow
The true power of TRLs and MMLs is realized when they are used synergistically throughout a research and development program. The MML tracks the maturation of the core material, ensuring that the fundamental building block is sufficiently understood and reliable, while the TRL tracks the integration of that material into a functional technology or product. A high MML de-risks the subsequent TRL progression. For instance, a new alloy for a surgical implant must first achieve a high MML (e.g., MML 6 or 7, demonstrating biocompatibility, mechanical strength, and fatigue resistance) before the surgical instrument itself can advance to higher TRLs (e.g., TRL 6 or 7, involving animal studies or clinical trials).
The following diagram illustrates the interconnected workflow between the materials tetrahedron, MMLs, and TRLs in a typical development process.
Experimental Protocols for Key Stages
To provide practical utility, below are generalized experimental protocols aligned with key transition points in the MML and TRL frameworks.
Protocol 1: MML 3 to MML 4 Transition (Establishing Processing-Property Relationships)
- Objective: To determine how a critical processing parameter (e.g., annealing temperature) affects a key material property (e.g., yield strength).
- Research Reagent Solutions:
- Material Samples: Raw material or precursor compounds.
- Processing Equipment: (e.g., Furnace with controlled atmosphere, sintering press, 3D printer).
- Characterization Tools: Scanning Electron Microscope (SEM) for microstructural analysis, X-ray Diffractometer (XRD) for phase identification.
- Property Testing Equipment: Universal Testing Machine (UTM) for tensile/compressive strength, Hardness Tester.
- Methodology:
- Sample Preparation: Prepare a series of material samples using a consistent base method but systematically varying the key processing parameter (e.g., create 5 batches annealed at 100°C intervals from 500°C to 900°C).
- Structural Characterization: Analyze the microstructure and phase composition of each sample using SEM and XRD.
- Property Testing: Measure the key property (e.g., yield strength) for each sample using the UTM according to relevant ASTM standards.
- Data Analysis: Plot the property value against the processing parameter. Use statistical analysis (e.g., regression) to establish a quantitative relationship and identify the optimal processing window.
Protocol 2: TRL 4 to TRL 5 Transition (Non-GLP In Vivo Demonstration for a Drug Candidate)
- Objective: To demonstrate preliminary proof-of-concept efficacy and safety of a candidate drug in a relevant animal model.
- Research Reagent Solutions:
- Test Article: The candidate drug substance, formulated for administration.
- Animal Model: A validated, biologically relevant species (e.g., mouse, rat, non-human primate).
- Assay Kits: ELISA kits for measuring biomarker levels, hematology analyzers.
- Histopathology Equipment: Tissue processor, embedder, microtome, staining apparatus.
- Methodology:
- Study Design: Define a dose-ranging study with a control group and at least three treatment groups receiving different doses of the candidate drug. Include a sufficient number of animals per group for statistical power.
- Dosing and Monitoring: Administer the drug according to the proposed clinical route (e.g., oral, intravenous). Monitor animals daily for clinical signs of toxicity and morbidity.
- Efficacy Endpoint Measurement: At defined timepoints, collect biological samples (e.g., blood, tissue) to measure predefined efficacy endpoints (e.g., reduction in pathogen load, tumor size reduction, biomarker modulation).
- Safety Evaluation: Conduct gross necropsy on all animals. Collect and preserve key organs for histopathological examination to identify any treatment-related lesions.
- Data Synthesis: Analyze efficacy and safety data to determine the dose-response relationship and identify a potentially safe and effective dose for further GLP studies.
The Impact of AI and Advanced Analytics on Readiness Acceleration
The application of Artificial Intelligence (AI) and Machine Learning (ML) is revolutionizing the pace at which materials and technologies advance through MML and TRL stages. In materials science, AI is being used as a "co-engineer" to learn from development data and autonomously recommend new material compositions or processing parameters, dramatically compressing the iterative cycle of the materials tetrahedron [3]. Projects like the TETRA initiative leverage AI and robotics to simultaneously explore composition and processing variants, achieving in days what traditionally took months [3]. In pharmaceuticals, AI is accelerating drug discovery by analyzing vast datasets to predict molecular interactions, optimize lead compounds, and even design clinical trials [71] [72]. For example, AI has been used to reduce the timeline for discovering a lead compound for fibrosis from years to under 18 months [72]. This AI-driven acceleration directly impacts readiness levels by providing high-fidelity predictions and automating experimental workflows, thereby de-risking the progression to higher MMLs and TRLs.
Technology Readiness Levels (TRLs) and Material Maturation Levels (MMLs) are complementary, powerful frameworks that provide much-needed structure and objectivity to the complex journeys of technology and material development. Rooted in the foundational principles of the materials tetrahedron, these frameworks enforce an evidence-based discipline that is critical for managing risk, allocating resources efficiently, and ultimately achieving success in highly competitive and regulated fields like pharmaceuticals and medical devices. The ongoing integration of AI and advanced analytics promises to further refine and accelerate these processes. For today's researchers, scientists, and drug development professionals, mastering the synergistic application of TRLs and MMLs is not merely an academic exercise but a strategic imperative for delivering innovative solutions to the market with greater speed, confidence, and efficacy.
The Processing-Structure-Property-Performance (PSPP) relationship, often visualized as the materials tetrahedron, represents a cornerstone of materials science and engineering. This framework establishes the critical causal links between how a material is made (processing), its resulting architecture at multiple length scales (structure), its measurable characteristics (properties), and its ultimate behavior in real-world applications (performance). A thorough understanding of PSPP linkages is vital for the rational design of new materials and the optimization of existing ones for advanced applications. This review provides a comprehensive comparative analysis of these foundational relationships across three major material classes: alloys, polymers, and ceramics. The paradigm of data-driven modeling, particularly machine learning (ML), is emerging as a powerful tool to decipher complex PSPP linkages, especially in additive manufacturing (AM) processes where relationships are highly non-linear [73]. This review synthesizes traditional knowledge with cutting-edge computational approaches to offer a unified perspective on PSPP relationships across material domains.
Theoretical Foundations of the PSPP Framework
The PSPP framework is a systematic approach for understanding the genesis of material behavior. Processing encompasses the synthesis and manufacturing steps, including parameters like temperature, pressure, and deformation paths. These processes dictate the material's Structure, which can be defined at multiple scales: atomic structure, crystal lattice, microstructural features (e.g., grain size, phase distribution), and meso/macro-structures (e.g., porosity, layers). The structure, in turn, determines the intrinsic and extrinsic Properties of the material, such as its mechanical, thermal, electrical, and chemical attributes. Finally, these properties collectively define the material's Performance in a specific service environment, such as its efficiency as a turbine blade, its durability as an implant, or its capacity in a battery.
The recent adoption of data-driven modeling has augmented traditional, often physics-based, approaches to establishing PSPP relationships. In additive manufacturing, for instance, ML models can serve as surrogates for costly physical simulations or experiments, automatically discovering patterns and trends in AM data to construct quantitative models of PSPP relationships across the parameter space [73]. This approach is particularly valuable for navigating the complex, multi-parameter design space presented by modern materials, including high-entropy alloys, polymer composites, and advanced technical ceramics.
PSPP Relationships in Alloys
Processing and Microstructural Development
The processing of alloys involves techniques such as casting, thermomechanical treatment, and increasingly, additive manufacturing. These processes profoundly influence the microstructural architecture. For instance, in advanced steels, processing conditions control phase transformations, leading to complex microstructures containing ferrite, austenite, bainite, and martensite in various proportions and morphologies [74]. The development of these structures is often studied through in-situ characterization methods, which provide deep insights into the kinetics and mechanisms of phase transformations [74]. In lightweight alloys (Al, Mg, Ti), processes like extrusion, deformation, and heat treatment are used to refine grain structure and control precipitation, directly impacting strength, ductility, and corrosion resistance [74]. Additive manufacturing introduces extremely high thermal gradients and solidification rates, resulting in non-equilibrium microstructures that deviate significantly from those produced by conventional means.
Structure-Property Linkages and Performance
The properties of alloys are a direct consequence of their microstructure. Key structure-property relationships include:
- Grain Size Strengthening: Hall-Petch relationship, where yield strength increases with decreasing grain size.
- Precipitation Hardening: Strength and hardness are enhanced by the presence of fine, coherent precipitates that impede dislocation motion.
- Transformation-Induced Plasticity (TRIP): In certain advanced steels, the strain-induced transformation of retained austenite to martensite enhances ductility and toughness.
The performance of alloys is evaluated based on application-specific metrics. For example, the performance of high-temperature alloys and intermetallics is gauged by their creep resistance and oxidation resistance in power generation and aerospace systems [74]. The drive towards improved efficiency in these sectors necessitates the development of alloys that can operate at ever-increasing temperatures, pushing the boundaries of material design [74].
Table 1: Key PSPP Linkages in Alloys
| Processing Method | Typical Microstructural Features | Resulting Properties | Exemplary Performance |
|---|---|---|---|
| Thermomechanical Processing | Fine grain size, dislocation networks | High strength, good toughness | Structural components in automotive/aircraft |
| Age Hardening | Coherent nano-scale precipitates | High yield strength, hardness | Aerospace frames, high-performance automotive |
| Additive Manufacturing | Non-equilibrium phases, fine cellular structures | Site-specific properties, potential anisotropy | Complex, lightweight geometries for aerospace |
| Quenching & Tempering | Tempered martensite | High strength-to-weight ratio | Tools, gears, and machinery components |
PSPP Relationships in Polymers
Processing and Structural Formation
Polymer processing, including techniques like injection molding, extrusion, and 3D printing, governs the chain orientation, crystallinity, and overall morphology. A prime example of intricate PSPP relationships is found in magnetically responsive polymer composites (MPCs) for soft robotics. The processing of these composites requires careful consideration of rheological properties to ensure uniform dispersion of magnetic particles (e.g., NdFeB, Fe₃O₄) within the polymer matrix (e.g., thermosets, thermoplastics) [9]. Sedimentation of microparticles can be prevented in high-viscosity systems, whereas nano-scale particles require stabilization strategies to avoid agglomeration [9]. Processing techniques range from simple mixing and solvent casting to more advanced methods like photolithography and 3D printing [9]. The application of magnetic fields during processing can be used to program magnetic anisotropy by inducing directional particle assembly, which is a clear example of processing directly dictating structure for a targeted property [9].
Property and Performance Outcomes
The properties of polymers and polymer composites are highly sensitive to their processed structure. For MPCs, the loading, distribution, and alignment of magnetic fillers define the composite's magnetic responsiveness, stiffness, and actuation behavior [9]. The thermal properties during processing are also critical; temperatures exceeding the Curie point (T_curie) of the filler can demagnetize it, while temperatures above the polymer's degradation temperature (T_d) can cause structural defects [9].
The performance of these materials is realized in their actuation capabilities. MPC-based robots can perform complex locomotion, including global translational motion (e.g., rolling, swimming) and shape-reconfigurable local motion (e.g., crawling, undulating) [9]. This performance is directly governed by the structural and physical characteristics of the MPCs, enabling applications in targeted drug delivery, minimally invasive surgery, and pollutant removal [9].
Table 2: Key PSPP Linkages in Polymer Composites
| Processing Method | Structural Features | Key Properties | Targeted Performance |
|---|---|---|---|
| Solvent Casting / Mixing | Isotropic particle distribution | Uniform magnetic response, baseline mechanical | Simple actuation, sensors |
| Field-Assisted Assembly | Anisotropic particle chains | Directional magnetic properties, tailored stiffness | Complex shape-morphing, targeted locomotion |
| Vat Photopolymerization | Complex 3D geometries, fine features | High resolution, tunable stiffness | Micro-robots, biomedical devices |
| Fused Deposition Modeling | Layered structure, potential anisotropy | Site-specific properties, rapid prototyping | Custom actuators, functional prototypes |
PSPP Relationships in Ceramics
Conventional and Additive Processing Routes
Ceramic processing has been revolutionized by additive manufacturing, which enables the production of complex geometries unattainable by conventional methods. Key AM techniques include Vat Photopolymerization (VPP) and Material Extrusion (MEX). These methods typically use composite materials, such as a photopolymer resin filled with ceramic particles (e.g., silica, alumina) or a ceramic-polymer filament, which are later debound and sintered at high temperatures to achieve a fully ceramic part [75]. The sintering process is a critical step that defines the final microstructure. Parameters like sintering temperature, holding time, and heating rate dramatically influence grain growth, pore size distribution, and densification, thereby dictating the final mechanical and thermal properties [76].
Microstructure, Properties, and High-Performance Applications
The structure of ceramics, particularly porosity, is a primary determinant of their properties. Research on SLA-printed silica ceramics has shown that optimizing sintering temperature and holding time can significantly enhance mechanical performance by reducing porosity variations [76]. For instance, a study comparing VPP and MEX manufactured alumina parts revealed distinct microstructures and properties. MEX specimens exhibited a more consistent microstructure before and after sintering, achieving a compressive strength of 168 MPa, whereas VPP specimens, despite having more repeatable results, reached a lower compressive strength of 81 MPa [75].
The performance of advanced ceramics is showcased in extreme environments. Ultra-High Temperature Ceramic Matrix Composites (UHTCMCs), composed of refractory carbides, borides, and nitrides, offer unparalleled thermal stability, oxidation resistance, and mechanical durability for aerospace propulsion and hypersonic vehicles [77]. Recent innovations, such as the development of a (Ti,Zr,Hf,Ta)CN/SiCN ceramic nanocomposite, demonstrate how precise molecular-level design and spark plasma sintering can yield materials with exceptional mechanical properties (flexural strength of 532–603 MPa) and outstanding ablation resistance, with a linear ablation rate two orders of magnitude lower than other UHTCMCs [78].
Table 3: Key PSPP Linkages in Advanced Ceramics
| Processing Method | Key Microstructural Traits | Resulting Properties | Performance in Application |
|---|---|---|---|
| Sintering (Conventional) | Grain size, density, pore morphology | Hardness, strength, thermal stability | Cutting tools, wear parts, substrates |
| Vat Photopolymerization | Complex geometry, fine features, layered structure | High design freedom, moderate strength | Prototypes, intricate filters, custom implants |
| Material Extrusion | Layered structure, potentially higher porosity | Variable strength, depends on infill | Porous structures, custom components |
| Spark Plasma Sintering | Fine grain size, high density, nanocomposites | Ultra-high strength, ablation resistance | Leading edges in hypersonic vehicles |
Comparative Analysis of PSPP Across Material Classes
A cross-comparison of PSPP relationships reveals fundamental similarities and critical distinctions among the three material classes. All three are profoundly influenced by processing parameters, but the underlying mechanisms differ. In metals, property control is predominantly through defect engineering (dislocations, grain boundaries). In polymers, it is governed by chain dynamics, cross-linking, and filler dispersion. In ceramics, the absence of dislocation-mediated plasticity at room temperature makes density, grain boundaries, and flaw population the dominant factors.
A unified challenge across all classes is the management of small data in machine learning for materials science. The acquisition of high-quality materials data often involves high experimental or computational costs, creating a dilemma between "simple analysis of big data and complex analysis of small data" [79]. Solutions are being developed at multiple levels: enriching data sources (e.g., database construction, high-throughput experiments), developing algorithms robust to small datasets, and employing strategic learning paradigms like active learning and transfer learning [79].
Experimental Protocols for PSPP Analysis
Protocol: PSPP Workflow for Additively Manufactured Ceramics
This protocol outlines the steps for establishing PSPP relationships in ceramics fabricated via Vat Photopolymerization, based on published methodologies [76].
Specimen Fabrication (Processing):
- Design: Create 3D models (e.g., cylinders for compression testing) and export as STL files.
- Slicing: Import STL files into CAM software. Set parameters including layer thickness (e.g., 0.05 mm or 0.10 mm), exposure time, and print speed.
- Printing: Manufacture "green" parts using a ceramic-loaded photopolymer resin on a VPP printer (e.g., Formlabs Form 2).
- Post-processing: Rinse parts in isopropyl alcohol to remove uncured resin and allow to dry.
Debinding and Sintering (Processing → Structure):
- Place green parts in a high-temperature furnace.
- Execute a controlled thermal cycle. For a silica-based ceramic, this involves:
- A slow ramp (e.g.,
1 °C/min) to600 °Cwith extended holds for polymer burnout (debinding). - A faster ramp (e.g.,
5 °C/min) to the peak sintering temperature (e.g.,1250 °C,1271 °C, or1300 °C). - Hold at peak temperature for a defined duration (e.g., 2 to 20 minutes).
- Cool down to room temperature at a controlled rate.
- A slow ramp (e.g.,
Microstructural Characterization (Structure):
- Section sintered specimens and prepare cross-sections using standard metallographic procedures (mounting, grinding, polishing).
- Perform quantitative microscopy (e.g., SEM) to analyze grain size, pore size distribution, and quantitative porosity.
- Use image analysis software to calculate area-weighted and number-weighted pore size distributions.
Mechanical Property Evaluation (Property):
- Conduct quasi-static uniaxial compression tests on sintered specimens.
- Record engineering stress-strain curves.
- Extract mechanical properties: compressive strength (
σ_c) and elastic modulus (E_c).
Data Correlation and Modeling (PSPP Linkage):
- Statistically correlate processing parameters (layer thickness, sintering temperature, hold time) with microstructural metrics (porosity, pore size) and mechanical properties (compressive strength).
- Use data-driven models (e.g., regression analysis, machine learning) to construct quantitative PSPP relationships.
The Scientist's Toolkit: Key Reagents and Materials
Table 4: Essential Research Reagents and Materials for PSPP Studies
| Item | Function in PSPP Research | Exemplary Use Case |
|---|---|---|
| Ceramic-Loaded Photopolymer Resin | Feedstock for creating complex "green" ceramic parts via VPP. | Formlabs Ceramic Resin for silica-based parts [76]. |
| High-Temperature Furnace | Enables debinding and sintering to remove polymer binder and densify the ceramic structure. | Used for thermal processing of printed ceramics [75]. |
| Magnetic Fillers (NdFeB, Fe₃O₄) | Provide magnetic responsiveness in a polymer composite matrix. | Functional filler in Magnetic Polymer Composites (MPCs) for soft robotics [9]. |
| Single-Source Precursors (SSPs) | Enable molecular-level design of complex ceramic compositions for superior homogeneity. | Synthesis of (Ti,Zr,Hf,Ta)CN/SiCN UHTCMCs [78]. |
| Spark Plasma Sintering (SPS) Setup | Advanced sintering technique using pulsed current for rapid densification of nanocomposites. | Fabrication of dense UHTCMCs with fine grain structure [78]. |
Visualization of PSPP Workflows
The following diagrams, generated using Graphviz, illustrate core PSPP concepts and workflows.
The Materials Tetrahedron and Data-Driven Modeling
Diagram 1: The core PSPP relationship and the role of data-driven modeling.
Detailed PSPP Workflow for Additive Manufacturing
Diagram 2: A detailed PSPP workflow for additive manufacturing across material classes.
This comparative analysis underscores that while the fundamental logic of the PSPP framework is universal across alloys, polymers, and ceramics, the specific mechanisms and controlling factors are distinct and deeply rooted in the inherent nature of each material class. The convergence of this foundational framework with modern data-driven approaches is creating a powerful paradigm for accelerating materials discovery and optimization. The ability of machine learning to navigate high-dimensional, non-linear PSPP relationships is particularly valuable for addressing the challenge of small data in materials science [79]. Future progress will depend on the continued integration of high-throughput experimentation, multi-scale modeling, and sophisticated data analytics to further elucidate and exploit the complex causal linkages that define material behavior from processing to performance.
The materials science tetrahedron is an enduring and powerful conceptual framework that captures the core philosophy of materials science and engineering. It illustrates the profound interdependence between four fundamental elements: processing, structure, properties, and performance [63] [11]. For decades, this paradigm has provided researchers with a systematic approach for understanding how materials are made, how they are structured, why they behave as they do, and how they ultimately function in real-world applications [80]. In the context of materials selection and benchmarking, the tetrahedron transitions from a mere educational diagram to an indispensable strategic tool. It offers a structured methodology for tracing the complex pathway from a material's synthesis conditions to its final application performance, enabling a more rigorous and predictive approach to selecting optimal materials for specific technological challenges [63]. This guide explores the practical application of the materials tetrahedron in benchmarking performance, with particular emphasis on its expanded role in the era of data-intensive science and its critical importance in fields such as pharmaceutical development.
Decoding the Tetrahedron: Core Components and Their Interrelationships
The Four Cornerstones
The four elements of the tetrahedron form a continuous cycle of cause and effect. A detailed understanding of each is prerequisite to applying the framework effectively.
- Processing encompasses the entire suite of techniques and conditions used to create, synthesize, or shape a material, from its raw constituents to a finished form [63]. This can range from primary processing (e.g., mining, purification, and alloying) to secondary processing (e.g., forging, heat treatment, and coating) [63]. In the pharmaceutical domain, this includes processes like crystallization, milling, and tablet compression [81].
- Structure refers to the arrangement of a material's internal components across multiple length scales. This includes atomic structure (bonding, crystal structure), microstructure (grains, phases, defects), and macroscopic structure [63] [80]. The classic example of diamond and graphite, both pure carbon with vastly different properties due to atomic structure, underscores its critical importance [63].
- Properties are the measurable responses of a material to specific external stimuli. These are the direct consequence of the material's structure and are typically categorized as mechanical, electrical, thermal, optical, or chemical [80]. Properties such as tensile strength, band gap, or dissolution rate are the metrics used for initial material screening.
- Performance describes how well a material fulfills its intended function in a final application or product [63]. This is the ultimate benchmark, encompassing not just intrinsic properties but also system-level factors like reliability, durability, and cost-effectiveness in a specific use case.
Visualizing the Workflow: From Processing to Performance
The following diagram maps the logical workflow of the materials tetrahedron, illustrating both the forward development path and the reverse materials selection pathway.
The Modern Tetrahedron: Integrating the Digital Twin and Data Science
The classic tetrahedron has evolved to embrace the digital transformation of materials science. The Materials-Information Twin Tetrahedra (MITT) framework augments the physical materials tetrahedron with a parallel "digital twin" representing its information science counterpart [1] [11]. This digital twin encompasses the data, models, and computational workflows that virtually represent the physical material throughout its life cycle.
The meta-framework of the MITT connects the materials domain to the information domain across six key dimensions, as summarized in the table below. This provides a systematic approach for benchmarking in a digital R&D environment.
Table: The Materials-Information Twin Tetrahedra (MITT) Meta-Framework
| Dimension | Materials Science Aspect | Information Science Aspect |
|---|---|---|
| Activities | Synthesis, characterization, testing | Inverse design, multiphysics simulation, autonomous experiments [1] |
| Arrangement | Structure, composition, phases | Digital representations, data structures, metadata schemas [1] |
| Qualities | Strength, conductivity, purity | Data accuracy, uncertainty, computational cost, bias [1] |
| Applicability | Suitability for a specific application | Clearly defined software scope and requirements [1] |
| Validation | Experimental verification | Benchmark datasets, objective metrics, validation of predictions [1] |
| Viability | Cost, supply risk, sustainability | FAIR data principles, sustained life cycle efficacy [1] |
This integrated framework is powered by the field of Materials Informatics (MI), which applies data-centric approaches, including machine learning (ML), to materials science R&D [82]. The strategic advantage of MI lies in its ability to accelerate the "forward" direction of innovation and, more powerfully, to enable the "inverse" direction—designing materials with desired properties from the outset [82].
Experimental Protocols for Tetrahedron-Guided Benchmarking
Applying the tetrahedron framework requires a disciplined experimental approach. The following workflow, applicable across diverse material classes, ensures a comprehensive and data-rich benchmarking process.
A Generalized Experimental Workflow
Key Methodologies and Research Reagents
The following table details essential reagents, tools, and methodologies that form the toolkit for executing this workflow, with examples from traditional and emerging material systems.
Table: Research Reagent Solutions for Tetrahedron-Guided Benchmarking
| Item Category | Specific Examples | Function in the Workflow |
|---|---|---|
| Processing Reagents | Basilisk self-healing agent (Bacillus bacteria species) [83]; PVDF polymer [83] | Creates the material; enables specific functionalities like autonomous crack repair or energy conversion. |
| Characterization Tools | X-ray Diffractometer (XRD); Scanning Electron Microscope (SEM); MRI with Metamaterial Enhancers [83] | Determines crystal structure, visualizes microstructure, and enhances signal-to-noise for superior imaging. |
| Property Testing Equipment | Universal Testing Machine; Impedance Analyzer; Dissolution Testing Apparatus | Quantifies mechanical strength, electrical properties, and dissolution kinetics for performance prediction. |
| Computational Resources | Open-source toolkits (e.g., AFLOW, Materials Project) [1]; Machine Learning Algorithms (e.g., Bayesian Optimization) [1] | Enables high-throughput virtual screening, predictive modeling, and inverse design of materials. |
Case Study: Benchmarking Self-Healing Concrete
- Performance Goal: Define metrics: maximum crack width healed, recovery of compressive strength, number of healing cycles [83].
- Processing: Incorporate bacteria (Bacillus subtilis, B. pseudofirmus, B. sphaericus) and nutrients into concrete mix design [83].
- Structure: Use SEM and micro-CT to characterize the distribution of bacterial capsules and the formation of new limestone (calcite) within cracks [83].
- Properties: Measure the recovery of mechanical properties (e.g., tensile and compressive strength) after cracking and healing cycles.
- Performance Benchmarking: Compare the durability and service life of the self-healing concrete against conventional concrete in accelerated aging tests.
Case Study: Benchmarking Metamaterials for 5G Antennas
- Performance Goal: Define metrics: antenna efficiency, bandwidth, signal-to-noise ratio in obstructed environments [83].
- Processing: Fabricate metamaterial unit cells using 3D printing, lithography, or etching with metals and dielectrics [83].
- Structure: Characterize the precise nanoscale architecture of the metamaterial using SEM and EDX to ensure it meets design specifications for electromagnetic response [83].
- Properties: Measure electromagnetic properties (e.g., negative refractive index, tailored permittivity) in an anechoic chamber.
- Performance Benchmarking: Integrate the metamaterial into an antenna prototype and test signal gain and reliability in a real-world urban environment [83].
Quantitative Benchmarking and Data Analysis
The final stage of the workflow involves the systematic comparison of data generated throughout the tetrahedron cycle. This is where the integration of the MITT framework becomes critical for modern research.
Benchmarking Data for Advanced Materials
The table below synthesizes illustrative data for emerging materials, demonstrating how the processing-structure-property-performance relationships can be quantified and compared.
Table: Benchmarking Data for Selected Advanced Materials
| Material & Application | Processing Parameter | Key Structural Feature | Measured Property | Performance Metric |
|---|---|---|---|---|
| Polymer Aerogel (Insulation) [83] | Drying method to create dendritic microstructure | Porosity >99.8%; pore size <100 nm | Thermal conductivity <0.02 W/m·K | Energy savings in buildings; SPF >50 in sunscreen [83] |
| Metamaterial (5G RIS) [83] | 3D printing/lithography | Sub-wavelength periodic architecture | Negative refractive index; beam-steering capability | +10 dB antenna gain; extended 5G range indoors [83] |
| Bamboo Composite (Sustainable Packaging) [83] | Plastination with polymers (e.g., silicone) | Bamboo fiber reinforcement in polymer matrix | Tensile strength >100 MPa; improved water vapor barrier [83] | 30% reduction in plastic use; biodegradable packaging |
| Electrochromic Window (Smart Building) [83] | Sputtering of tungsten trioxide / nickel oxide films | Multi-layer thin-film stack | Optical switching speed <3 minutes; >60% transparency change [83] | ~20% reduction in building HVAC energy use [83] |
The Role of FAIR Data and Informatics in Benchmarking
Effective benchmarking in the 21st century relies on the principles of Findable, Accessible, Interoperable, and Reusable (FAIR) data [1] [11]. By ensuring data generated at each corner of the tetrahedron adheres to these principles, researchers can:
- Build high-quality, large-scale datasets for machine learning.
- Directly compare results across different studies and laboratories.
- Accelerate the discovery of new processing-structure-property-performance relationships.
The application of materials informatics tools allows researchers to move beyond simple comparison. By using the benchmarking data to train ML models, one can predict the performance of untested materials or optimize processing parameters to achieve a performance goal, thereby closing the loop on the tetrahedron and realizing the full potential of the inverse design paradigm [82].
The materials science tetrahedron remains a vital framework for guiding the complex process of materials selection and performance benchmarking. Its power lies in providing a systematic, holistic view of the cause-and-effect relationships that define a material's journey from synthesis to application. The modern extension of this framework through the Materials-Information Twin Tetrahedra (MITT) and the tools of materials informatics has transformed it from a primarily descriptive model into a dynamic, predictive, and iterative engine for innovation. For researchers and drug development professionals, rigorously applying this integrated framework ensures that materials selection is no longer a matter of educated guesswork, but a data-driven, strategic process capable of meeting the most demanding application challenges.
The materials science tetrahedron (MST)—encompassing the interdependent relationship between a material's processing, structure, properties, and performance—provides a foundational framework for materials design. The vast majority of data required to populate this framework is locked within unstructured text, tables, and images of published scientific literature. This whitepaper examines the role of advanced information extraction (IE) techniques, particularly those leveraging large language models (LLMs), as a critical tool for validating and reconstructing the materials tetrahedron from this dispersed data. We explore the significant machine learning challenges inherent in this process, quantify the distribution of key material data within research papers, and present detailed protocols for automated data extraction. By transforming published literature into a structured, queryable knowledge base, IE enables the validation of material behavior hypotheses and accelerates the discovery and development of new materials, including pharmaceuticals, with unprecedented efficiency.
The materials science tetrahedron (MST) is a cornerstone concept in materials engineering, concisely depicting the interdependent relationship among a material's structure, properties, performance, and processing [2]. This framework is equally vital in pharmaceutical materials science, where the performance of a drug product is directly linked to the active pharmaceutical ingredient's (API) solid-state structure and the processing methods used to formulate it [2]. Understanding a material's behavior requires synthesizing knowledge across all four of these components.
However, this information is reported across millions of scientific documents in a myriad of unstructured or semi-structured formats, including peer-reviewed articles, books, and patents. There is little to no uniformity in reporting style, creating a significant bottleneck for the development of a comprehensive materials knowledge base [18]. Information extraction (IE) has emerged as a set of computational techniques to overcome this bottleneck. By automatically identifying and retrieving specific data points—such as material compositions, synthesis parameters, and property measurements—from literature, IE serves as a powerful means of validating the relationships postulated by the tetrahedron. The process of extracting and linking this data allows researchers to test hypotheses on a vast scale, confirm or challenge existing models, and ultimately, reconstruct the tetrahedron from published evidence.
The Information Landscape in Materials Science Literature
A quantitative analysis of where key tetrahedral components are reported is essential for designing effective IE systems. A study of 2,536 materials science publications revealed the distribution of information across texts and tables, as summarized in Table 1 [18].
Table 1: Prevalence of Materials Tetrahedron Components in Scientific Literature
| Tetrahedron Component | Reported in Text | Reported in Tables | Key Finding |
|---|---|---|---|
| Material Compositions | 78% of papers | 74% of papers | 85.92% of all compositions were found in tables. |
| Material Properties | Information not specified | 82% of papers | The vast majority of properties are tabulated. |
| Processing Conditions | Mostly in text | Information not specified | Details are primarily found in the experimental sections of text. |
| Testing Conditions | Mostly in text | Information not specified | Details are primarily found in the experimental sections of text. |
| Precursors (Raw Materials) | 80% of papers | Information not specified | Predominantly reported in textual descriptions. |
This distribution highlights a critical insight: while compositions and properties are predominantly found in tables, processing and testing conditions are largely described in prose. Furthermore, an in-depth analysis of compositions revealed that although a high percentage of papers mention compositions in text, the vast majority of actual compositional data points (85.92%) reside in tables [18]. This makes tables a particularly rich target for IE efforts aimed at completing the materials tetrahedron.
Core Challenges in Materials Information Extraction
Automated IE from materials science literature faces several interconnected challenges, which are quantified in Table 2 below. These challenges directly impact the ability to accurately reconstruct the materials tetrahedron.
Table 2: Key Challenges in Extracting Material Compositions from Tables
| Challenge Category | Specific Challenge | Frequency of Occurrence | Impact on IE |
|---|---|---|---|
| Table Structure | Multi-Cell Composition (MCC) Tables | 36% (MCC-CI) | Requires parsing data across multiple cells/columns. |
| Single-Cell Composition (SCC) Tables | 30% (SCC-CI) | Requires parsing a string of text within a single cell. | |
| Partial Information (PI) Tables | 34% (MCC-PI & SCC-PI) | Necessitates cross-referencing with the article's text. | |
| Data Reporting | Nominal vs. Experimental Compositions | 3% of tables | Difficult to distinguish between intended and actual composition. |
| Use of Material IDs/References | 10% of tables | Data is incomplete without consulting external cited documents. | |
| Entity Linking | Connecting data across text, tables, and images | Pervasive | Required to form complete tetrahedron data points (e.g., linking a property in a table to its processing condition in the text). |
The variation in table structure is a primary obstacle. Tables can be categorized as Single-Cell Composition (SCC), where the entire composition is written inside one cell, or Multi-Cell Composition (MCC), where the value of each constituent is reported in separate cells. Each type can contain either Complete Information (CI) or only Partial Information (PI), the latter requiring the IE system to find the missing data in the surrounding text [18]. This lack of a standard schema makes it difficult to develop a one-size-fits-all extraction model.
Other complications include the presence of both nominal (the amount of chemicals taken initially) and experimental/analyzed (the actual composition of the manufactured material) compositions in the same table, reported without a fixed pattern [18]. Furthermore, about 10% of composition tables use material IDs or references to prior work instead of explicitly stating the composition, forcing an IE system to look beyond the immediate document [18]. The ultimate challenge is entity linking—connecting the extracted information on composition, structure, properties, and processing that is spread across different formats and sections of a paper to form a coherent data point for the materials tetrahedron [84] [18].
Methodologies for Data Extraction and Validation
The ChatExtract Protocol for Data Extraction from Text
The ChatExtract methodology represents a state-of-the-art approach for automating the extraction of structured data (e.g., Material, Value, Unit triplets) from unstructured text using conversational large language models (LLMs) in a zero-shot fashion (i.e., without additional model training) [85]. Its high accuracy (precision and recall both close to 90% with GPT-4) makes it highly suitable for scientific data mining. The workflow, illustrated in Figure 1, is as follows:
Figure 1: The ChatExtract Workflow for Automated Data Extraction
Key Experimental Steps and Prompt Engineering Features:
- Initial Relevancy Classification (Stage A): Every sentence is processed with a simple prompt to determine if it contains relevant data (a value and unit). This efficiently weeds out the ~99% of sentences that are irrelevant [85].
- Passage Expansion: For sentences classified as relevant, the text passage is expanded to include the paper's title and the preceding sentence. This ensures the material's name, which is often not in the target sentence itself, is included for context [85].
- Single vs. Multi-Valued Data Paths (Stage B): The system differentiates between sentences containing a single data value and those with multiple values. This is crucial because extraction from multi-valued texts is more prone to errors and requires more rigorous verification [85].
- Uncertainty-Inducing Redundant Prompts: This is the core innovation for ensuring accuracy. For complex, multi-valued sentences, the LLM is asked a series of follow-up questions that suggest uncertainty about its initial extraction (e.g., "Are you sure that value corresponds to that material?"). This prompts the model to re-analyze the text instead of reinforcing a potentially incorrect initial answer, largely overcoming the issue of LLM "hallucinations" [85].
- Strict Yes/No Answer Enforcement: Follow-up questions are designed to be answered with a strict "Yes" or "No," reducing ambiguity and simplifying automated processing [85].
The TETRA Protocol for Integrated Physical and Data Workflows
While ChatExtract focuses on extracting knowledge from existing literature, the TETRA (Transforming Evaluation and Testing via Robotics and Acceleration) initiative exemplifies a parallel, forward-looking paradigm that generates new high-throughput experimental data, thereby creating a well-structured tetrahedron from the outset [3]. This integrated physical and data workflow is depicted in Figure 2.
Figure 2: The TETRA Integrated Physical and Data Workflow
Key Experimental Steps and Components:
- Combinatorial Synthesis via Additive Manufacturing: Instead of producing one large ingot of a single composition, TETRA uses techniques like blown-powder directed energy deposition (DED). A laser melts metal powder as it's fed into a build area, allowing for the creation of a single build plate containing hundreds of different alloys, printed as custom-designed 3D specimens ready for testing [3].
- Automated Processing: The synthesized samples are then processed using custom, automated furnaces for heat treatment and robotic forging equipment. This is critical as it accounts for the effects of processing on microstructure and properties, a key leg of the materials tetrahedron often missing from purely computational screening [3].
- Robotic Mechanical Property Measurement: Robotic systems autonomously conduct mechanical tests on the hundreds of processed specimens. This generates consistent, high-quality property data at an unprecedented rate [3].
- AI Co-Investigator and Closed Loop: The structured data generated from synthesis, processing, and testing is fed to an AI. The AI learns from these results and automatically recommends the next set of experiments to run, or even designs new materials, creating a closed-loop, autonomous materials discovery and optimization system [3].
The Scientist's Toolkit: Research Reagent Solutions
The following table details key computational and experimental tools essential for implementing the IE and validation workflows described in this whitepaper.
Table 3: Essential Tools for Tetrahedron Data Extraction and Generation
| Tool Name / Category | Type / Function | Brief Description of Role |
|---|---|---|
| Conversational LLMs (e.g., GPT-4) | Computational / Data Extraction | The core engine for zero-shot IE protocols like ChatExtract; used to identify and parse material data from text with high accuracy [85]. |
| Directed Energy Deposition (DED) | Experimental / Synthesis | An additive manufacturing technique used in TETRA for combinatorial synthesis, enabling rapid fabrication of many alloy variants on a single build plate [3]. |
| Custom Robotic Test Rigs | Experimental / Characterization | Automated systems for performing mechanical and property tests on synthesized samples, enabling high-throughput data generation for the properties-performance tetrahedron link [3]. |
| DiSCoMaT | Computational / Table Extraction | A domain-specific IE model designed to extract material compositions from scientific tables, though it struggles with partial information tables and material IDs [18]. |
| ChartExpo | Computational / Data Visualization | A tool for creating advanced visualizations to communicate the quantitative relationships (e.g., property vs. structure) uncovered through IE and high-throughput experimentation [86]. |
The reconstruction of the materials tetrahedron from published data through advanced information extraction is no longer a theoretical pursuit but an emerging reality. Techniques like the ChatExtract protocol demonstrate that conversational LLMs can achieve high-precision, high-recall data extraction with minimal upfront effort, effectively converting unstructured literature into a structured knowledge base. Concurrently, initiatives like TETRA are creating a new paradigm where the tetrahedron is natively built from structured, high-throughput experimental data, with AI acting as a co-investigator.
The future of materials development lies in the synergy of these two approaches. As LLMs and IE techniques become more sophisticated, they will allow us to mine the vast, untapped knowledge of the past century of materials research. Simultaneously, autonomous and high-throughput labs will systematically generate the data for the next century. Integrating these vast, structured knowledge bases will enable truly predictive materials science, where the relationships of the tetrahedron can be modeled, validated, and exploited with unparalleled speed and accuracy, profoundly impacting fields from aerospace to pharmaceutical development.
The foundational paradigm in materials science and engineering is the materials tetrahedron, which illustrates the intricate interrelationships between processing, structure, properties, and performance. Traditionally, navigating these relationships to develop new materials has been a time-consuming, iterative process heavily reliant on researcher intuition and sequential experimentation. However, the emergence of artificial intelligence (AI) and machine learning (ML) is fundamentally transforming this workflow, enabling a more integrated and accelerated approach to discovery [87]. This shift represents a transition from a human-led, linear process to a data-driven, cyclic one where AI assists in planning experiments, analyzing results, and refining hypotheses.
This technical guide provides an in-depth comparison of traditional and AI-accelerated materials development workflows. It examines their underlying methodologies, experimental protocols, and practical implementations through real-world case studies, framed within the context of the materials tetrahedron. The analysis is particularly relevant for researchers and professionals engaged in the development of advanced functional materials for applications in energy, electronics, and nanotechnology.
Traditional Materials Development Workflow
Core Principles and Methodology
The traditional materials development workflow is characterized by its sequential, human-centric, and experience-driven nature. This approach relies heavily on established scientific principles, researcher intuition honed through hands-on work, and methodical experimental iteration [88]. The process typically follows a linear path through the materials tetrahedron, with decisions based on prior knowledge and phenomenological understanding.
The traditional workflow depends substantially on high-throughput experimental (HTE) approaches to explore material libraries. At institutions like NREL, this involves combinatorial deposition systems that create intentional, well-controlled gradients in chemical composition, substrate temperature, and film thickness across a substrate [89]. This generates numerous material variations in a single batch, which are then characterized using automated measurement instruments with controlled motion stages to map properties as a function of position.
Key Experimental Protocols
Synthesis and Processing Methods
Traditional synthesis employs well-established physical and chemical methods:
- Physical Vapor Deposition (PVD): Creating thin films through controlled vaporization and condensation of precursor materials in vacuum chambers [89].
- Sol-Gel Processing: Producing solid materials from small molecules in solution, enabling precise control over composition and structure at low temperatures [90].
- Solid-State Reaction: Processing bulk materials through high-temperature treatment of powder mixtures to facilitate atomic diffusion and reaction.
- Hydrothermal Synthesis: Conducting crystallization in aqueous solutions at elevated temperature and pressure to create structured materials.
Characterization and Analysis
Material characterization in traditional workflows relies on well-established techniques:
- X-ray Diffraction (XRD): Analyzing crystal structure and phase composition through diffraction patterns.
- Scanning Electron Microscopy (SEM): Investigating microstructural features and morphology with high resolution.
- Automated Property Mapping: Using motion-controlled stages to systematically measure properties across combinatorial libraries [89].
Data Analysis Approach
Data analysis in traditional workflows involves:
- Statistical Analysis: Applying conventional statistical methods to identify correlations and trends.
- Expert Interpretation: Relying on researcher experience and domain knowledge to draw conclusions from experimental data.
- Sequential Refinement: Using results from one experiment to inform the next iteration in a linear fashion.
Table 1: Key Reagents and Equipment in Traditional Materials Development
| Category | Specific Examples | Function in Workflow |
|---|---|---|
| Synthesis Equipment | Physical Vapor Deposition Chambers | Create controlled material libraries with compositional gradients [89] |
| Carbothermal Shock Systems | Rapid synthesis of materials through extreme thermal processing | |
| Characterization Tools | Automated XRD with X-Y Stages | Structural analysis across material libraries [89] |
| Scanning Electron Microscopy | Microstructural imaging and analysis | |
| Precursor Materials | Metal Salts, Organometallics | Source materials for thin film deposition and bulk synthesis |
| Polymer Solutions | Base materials for polymer-based composites and aerogels |
Limitations and Challenges
The traditional approach faces several significant limitations:
- Temporal Inefficiency: The sequential nature of experimentation creates lengthy development cycles, often taking years to advance from concept to viable material [87].
- Combinatorial Complexity: The vastness of possible chemical compositions and processing parameters makes comprehensive exploration impractical [88].
- Human Cognitive Constraints: Researchers' ability to identify complex, multi-variable relationships from experimental data is inherently limited.
- Reproducibility Issues: Subtle variations in processing conditions can significantly impact material properties, creating challenges in experimental consistency [91].
AI-Accelerated Materials Development Workflow
Core Principles and Methodology
AI-accelerated materials development represents a paradigm shift from sequential experimentation to an integrated, data-driven approach. This methodology leverages machine learning algorithms to rapidly navigate the complex relationships within the materials tetrahedron, enabling predictive design and optimized synthesis parameters [87]. Instead of relying solely on human intuition, these systems learn from diverse data sources - including experimental results, scientific literature, and computational simulations - to suggest promising research directions.
Central to this approach is the concept of active learning, where AI systems not only analyze existing data but also proactively suggest which experiments to perform next to maximize information gain [91]. As explained by MIT researchers, "Bayesian optimization is like Netflix recommending the next movie to watch based on your viewing history, except instead it recommends the next experiment to do" [91]. This creates a cyclic workflow where each experiment informs the next in an optimized sequence.
Key AI Technologies and Implementation
Machine Learning Approaches
- Bayesian Optimization (BO): Guides experimental design by balancing exploration of new parameter spaces with exploitation of known promising regions [91] [87].
- Graph Neural Networks (GNNs): Effectively represent crystal structures and molecular arrangements as graphs, enabling accurate property predictions [87].
- Generative Models: Including Generative Adversarial Networks (GANs) and Variational Autoencoders (VAEs) that can propose novel chemical compositions meeting specific criteria [87].
- Gaussian Process Models: Used in frameworks like ME-AI (Materials Expert-Artificial Intelligence) to uncover quantitative descriptors from expert-curated data [88].
Autonomous Experimentation Systems
Advanced platforms like MIT's CRESt (Copilot for Real-world Experimental Scientists) integrate robotic equipment with AI guidance [91]. These systems feature:
- Liquid-handling robots for precise sample preparation
- Automated synthesis systems like carbothermal shock for rapid material creation
- High-throughput characterization including automated electron microscopy
- Computer vision monitoring to detect issues and suggest corrections
Multimodal Data Integration
Unlike traditional methods that often rely on single data streams, AI-accelerated workflows incorporate diverse information types:
- Scientific literature insights and historical data
- Chemical composition and structural information
- Microstructural images from various microscopy techniques
- Experimental results from both successful and failed attempts
- Human feedback and researcher intuition [91]
Experimental Protocols in AI-Accelerated Workflows
Data Curation and Feature Engineering
The ME-AI framework demonstrates the importance of careful data curation, where experts select experimentally accessible primary features based on chemical intuition and domain knowledge [88]. For square-net compounds, these might include:
- Atomistic features: Electron affinity, electronegativity, valence electron count
- Structural features: Crystallographic distances (e.g., square-net distance dsq, nearest-neighbor distance dnn)
- Elemental properties: Estimated lattice parameters of constituent elements
Autonomous Synthesis and Characterization
AI-accelerated systems implement automated experimental protocols:
- Recipe Optimization: AI suggests material compositions and processing parameters
- Robotic Synthesis: Automated systems prepare samples according to optimized recipes
- High-Throughput Characterization: Multiple properties are measured in parallel
- Data Integration: Results are fed back into ML models to refine predictions
- Iterative Optimization: The cycle repeats with progressively improved materials
Active Learning Loops
The CRESt platform exemplifies the active learning approach [91]:
- Uses previous literature and databases to create representations of recipes
- Performs principal component analysis to identify reduced search spaces
- Applies Bayesian optimization in this reduced space to design new experiments
- Incorporates newly acquired multimodal data and human feedback to augment knowledge
- Continuously refines the search space to boost learning efficiency
Table 2: Key AI Technologies and Their Applications in Materials Development
| AI Technology | Specific Function | Impact on Workflow |
|---|---|---|
| Bayesian Optimization | Recommends next experiments based on all previous data [91] | Reduces number of experiments needed by focusing on most promising candidates |
| Graph Neural Networks | Represents complex material structures as graphs for property prediction [87] | Enables accurate prediction of properties without synthesis |
| Generative Models | Proposes novel chemical compositions meeting specific criteria [87] | Expands search beyond known chemical spaces to discover new materials |
| Computer Vision | Monitors experiments visually and detects issues [91] | Improves reproducibility by identifying subtle experimental variations |
| Large Language Models | Extracts knowledge from scientific literature and enables natural language interaction [91] | Makes system accessible to researchers without coding expertise |
Comparative Analysis: Workflow Efficiency and Outcomes
Quantitative Performance Metrics
The differences between traditional and AI-accelerated workflows become evident when examining specific performance metrics:
Table 3: Workflow Comparison - Traditional vs. AI-Accelerated Approaches
| Performance Metric | Traditional Workflow | AI-Accelerated Workflow | Comparative Advantage |
|---|---|---|---|
| Exploration Scale | Limited by human capacity and resource constraints | 900+ chemistries explored in 3 months (CRESt case) [91] | 10x-100x greater exploration efficiency |
| Experimental Cycle Time | Weeks to months per iteration | Hours to days per iteration with robotic systems | 5x-10x faster iteration cycles |
| Resource Utilization | High per data point, with significant material waste | Optimized to maximize information per experiment | 30-50% reduction in experimental costs [82] |
| Success Rate | Low (0.1-1%) in uncharted chemical spaces | Higher (5-10%) through targeted suggestions | 5x-10x improvement in discovery rate |
| Human Involvement | Hands-on throughout process | Oversight and strategic guidance | Researchers focus on high-value decisions |
| Reproducibility | Variable due to manual processes | Enhanced through automated monitoring and control [91] | Significant improvement in experimental consistency |
Case Study: Fuel Cell Catalyst Development
A direct comparison emerges from catalyst development for direct formate fuel cells:
In the traditional approach, researchers would typically:
- Select catalyst candidates based on literature and periodic table trends
- Manually synthesize small batches of promising compositions
- Characterize structure and test performance sequentially
- Iterate slowly based on results, often focusing on pure metals or simple alloys
The AI-accelerated approach using the CRESt platform demonstrated [91]:
- Exploration of 900+ chemistries over three months
- Conduct of 3,500+ electrochemical tests
- Discovery of an 8-element catalyst with record power density
- Achievement of 9.3-fold improvement in power density per dollar over pure palladium
- Operation with one-fourth the precious metals of previous devices
This case illustrates how AI-accelerated workflows can navigate complex multi-element composition spaces that would be practically infeasible through traditional methods.
Integrated Workflow Visualization
The fundamental difference between the two approaches can be visualized as a transformation from a linear, human-centric process to a cyclic, AI-integrated system:
Diagram 1: Workflow Architecture Comparison. The AI-accelerated approach introduces critical feedback loops that enable continuous optimization, contrasting with the linear traditional process.
The Scientist's Toolkit: Essential Research Reagents and Solutions
Table 4: Key Research Reagents and Solutions in AI-Accelerated Materials Development
| Category | Specific Examples | Function in Workflow |
|---|---|---|
| AI/ML Platforms | CRESt (MIT) [91], ME-AI [88], AutoGluon, TPOT [87] | Provide active learning, experiment planning, and data analysis capabilities |
| Robotic Equipment | Liquid-handling Robots, Automated Electrochemical Workstations [91] | Enable high-throughput synthesis and characterization without manual intervention |
| Characterization Tools | Automated Electron Microscopy, X-ray Diffraction Systems [91] [89] | Provide structural and compositional data for ML model training |
| Data Management | Materials Project, OQMD, AFLOW, NOMAD Databases [87] | Supply training data from computational and experimental sources |
| Computational Resources | GPU/TPU Clusters, Cloud Computing Platforms [87] | Accelerate model training and quantum calculations for material screening |
The comparison between traditional and AI-accelerated materials development workflows reveals a fundamental transformation in how researchers navigate the materials tetrahedron. While traditional methods rely on sequential experimentation guided by human intuition, AI-accelerated approaches leverage machine learning to create integrated, data-driven discovery cycles. The case studies demonstrate that AI-accelerated workflows can achieve order-of-magnitude improvements in exploration efficiency, success rates, and development timelines.
Looking forward, several trends are poised to further accelerate this transformation. The development of foundation models for materials science will enable more accurate predictions across diverse material classes [82]. Increased integration of autonomous robotic systems will close the loop between computation, synthesis, and characterization [91] [87]. Furthermore, approaches that better incorporate human expertise with AI capabilities, as demonstrated by the ME-AI framework [88], will create more effective human-AI collaborative systems.
For researchers and drug development professionals, these advances highlight the growing importance of developing skills in data science and machine learning alongside traditional materials expertise. The future of materials development lies not in replacing researchers, but in augmenting their capabilities with AI systems that can navigate complex multidimensional spaces more efficiently than humans alone. As these technologies mature, they promise to dramatically accelerate the discovery and development of advanced materials addressing critical challenges in energy, healthcare, and sustainability.
Conclusion
The Materials Science Tetrahedron remains an indispensable framework for navigating the complex, interdependent landscape of material design, proving particularly vital for the precise demands of pharmaceutical development. By systematically applying the Processing-Structure-Properties-Performance relationships, researchers can transition from serendipitous discovery to rational, efficient design of advanced drug delivery systems and biomaterials. Future directions point toward an increasingly digital paradigm, where AI co-investigators, self-driving labs, and robust knowledge bases will dramatically compress development timelines. For the biomedical field, this evolution promises a new era of tailored materials that meet stringent requirements for efficacy, safety, and manufacturability, ultimately accelerating the delivery of groundbreaking therapies to patients.
References
- 1. The materials tetrahedron has a “digital twin” - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9365726/]
- 2. Materials Science Tetrahedron—A Useful Tool for ... [https://www.sciencedirect.com/science/article/abs/pii/S0022354916329616]
- 3. An Accelerated Paradigm for Developing Mission-Critical ... [https://www.jhuapl.edu/news/news-releases/250917-tetra-mission-critical-materials]
- 4. Materials designed to degrade: structure, properties ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11498330/]
- 5. Revolutionizing Materials Science and Technology to ... [https://www.nae.edu/341005/Revolutionizing-Materials-Science-and-Technology-to-Secure-Our-Future]
- 6. On the importance of microstructure information in ... [https://www.sciencedirect.com/science/article/abs/pii/S1359645421008508]
- 7. Materials designed to degrade: structure, properties ... [https://pubs.rsc.org/en/content/articlelanding/2025/py/d4py00623b]
- 8. PROSPECT-PSPP: an automatic computational pipeline for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC441552/]
- 9. Processing–Structure–Property–Performance relationships ... [https://www.sciencedirect.com/science/article/abs/pii/S007967002500084X]
- 10. Establishing a process-structure-property-performance ... [https://www.sciencedirect.com/science/article/pii/S0264127525008512]
- 11. The materials tetrahedron has a “digital twin” | MRS Bulletin [https://link.springer.com/article/10.1557/s43577-021-00214-0]
- 12. A comprehensive review on the rational design of MOF ... [https://www.sciencedirect.com/science/article/abs/pii/S0277538725001846]
- 13. Materials designed to degrade: structure, properties ... [https://pubs.rsc.org/en/content/articlehtml/2025/py/d4py00623b]
- 14. Industry 4.0 & the Future of the Pharmaceutical Industry [https://ispe.org/pharmaceutical-engineering/march-april-2021/industry-40-future-pharmaceutical-industry]
- 15. New Drug Manufacturing Plants: A 2025 Guide & Analysis [https://intuitionlabs.ai/articles/new-drug-manufacturing-plants-2025]
- 16. Conceptualizing the research cycle for the field of materials ... [https://link.springer.com/article/10.1557/s43577-025-00931-w]
- 17. Processing–Structure–Properties Relationships of ... [https://www.mdpi.com/journal/materials/special_issues/9A673SL95M]
- 18. challenges in materials information extraction - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2024/dd/d4dd00032c]
- 19. The materials tetrahedron has a "digital twin". - Scholars@Duke [https://scholars.duke.edu/individual/pub1509623]
- 20. Data-driven modeling of process-structure-property ... [https://www.nature.com/articles/s44334-024-00003-y]
- 21. An integrated approach combining experimental ... [https://www.sciencedirect.com/science/article/abs/pii/S0022354924004477]
- 22. Current situation and challenges of polyhydroxyalkanoates ... [https://www.sciencedirect.com/science/article/pii/S259018342400053X]
- 23. Separation and purification technologies in ... [https://www.eeer.org/journal/view.php?number=1649]
- 24. Fabrication and Evaluation of Polyhydroxyalkanoate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11944280/]
- 25. Polyhydroxyalkanoates for sustainable food packaging [https://www.sciencedirect.com/science/article/abs/pii/S0924224425004303]
- 26. Recent Advances in Magnetic Polymer Composites for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10223786/]
- 27. Magnetic Polymer Composite Particles: Design and ... [https://www.mdpi.com/2073-4360/13/4/512]
- 28. Eco-sustainable magnetic polymer composites using recycled ... [https://pubs.rsc.org/en/content/articlehtml/2025/su/d5su00222b]
- 29. Fabrication and Applications of Magnetic Polymer ... [https://www.mdpi.com/2072-666X/14/12/2173]
- 30. Synthesis and Characterization of Magnetic Molecularly ... [https://www.mdpi.com/1422-0067/26/16/7686]
- 31. PVA composites in various structural forms: compact films ... [https://pubs.rsc.org/en/content/articlehtml/2025/nr/d5nr00450k]
- 32. Heilmeier Catechism: A DARPA's Way to Fund Projects [https://www.greyb.com/blog/heilmeier-catechism-a-darpas-way-to-fund-projects/]
- 33. Program Manager Application [https://arpa-h.gov/careers/program-managers]
- 34. The Heilmeier Catechism [https://lab.abilian.com/Business/R%26D%20and%20Innovation/The%20Heilmeier%20Catechism/]
- 35. Creating Impactful One-Pagers Using the Heilmeier Catechism [https://techtransferfordefense.stanford.edu/creating-impactful-one-pagers-using-heilmeier-catechism]
- 36. Crafting Better Proposals Using the Heilmeier Catechism [https://www.depts.ttu.edu/research/ordc/Resources/heilmeier-catechism.php]
- 37. Technology Transfer for Defense: Leveraging the Heilmeier ... [https://stanfordh4d.substack.com/p/technology-transfer-for-defense-leveraging]
- 38. Effectively Framing Your Research in an Increasingly ... [https://case.edu/research/about/ortm-updates/effectively-framing-your-research-increasingly-complex-funding-environment]
- 39. Preparing White Papers and Quad Charts [https://www.umkc.edu/osi/funding-announcements/preparing-white-papers-and-quad-charts.html]
- 40. Does The Heilmeier Catechism Need A 2.0? Reflections ... [https://the-learning-agency.com/insights/does-the-heilmeier-catechism-need-a-2-0-reflections-from-arpa-experts/]
- 41. RIO ARPA-H Info & Assistance | Research & Innovation Office [https://www.colorado.edu/researchinnovation/research-development/funding/external-funding/rio-arpa-h-info-assistance]
- 42. New insight into the multivariate relationships among ... [https://www.sciencedirect.com/science/article/abs/pii/S2214860423004177]
- 43. A Case Study of Multimodal, Multi-institutional Data ... [https://link.springer.com/article/10.1007/s40192-024-00345-7]
- 44. Experimental protocols and models for assessing the ... [https://www.sciencedirect.com/science/article/pii/S0304389425018497]
- 45. SPIRIT 2025 statement: Updated guideline for protocols of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12037212/]
- 46. Next-Generation Food Packaging: Progress and Challenges of... [https://pubmed.ncbi.nlm.nih.gov/40942216/]
- 47. Biopolymer Production Cost Analysis 2025 | Plant Setup [https://www.imarcgroup.com/biopolymer-manufacturing-plant-project-report]
- 48. The Future of Biopolymers [https://www.numberanalytics.com/blog/future-biopolymers-trends-challenges-opportunities]
- 49. Leadvent Group| Biopolymers , Plastics Industry, Sustainable... [https://www.leadventgrp.com/blog/the-rise-of-biopolymers-revolutionizing-the-plastics-industry]
- 50. Editorial: Journal of Materials Science: Metallurgy [https://link.springer.com/article/10.1007/s44492-025-00001-x]
- 51. Biopolymer Production Business Plan 2025 | CapEx, OpEx ... [https://www.linkedin.com/pulse/biopolymer-production-business-plan-2025-capex-opex-roi-adhikari-dn69e]
- 52. How Soybean-Based Biopolymer Works — In One Simple Flow ( 2025 ) [https://www.linkedin.com/pulse/how-soybean-based-biopolymer-works-one-simple-flow-2025-marketmorph-zqp0e]
- 53. Microfluidic systems and ultrasonics for emulsion-based biopolymers ... [https://researchportal.hw.ac.uk/en/publications/microfluidic-systems-and-ultrasonics-for-emulsion-based-biopolyme]
- 54. LLM-Based Information Extraction to Support Scientific ... [https://arxiv.org/html/2510.04749v1]
- 55. A review on knowledge and information extraction from ... [https://www.frontiersin.org/journals/artificial-intelligence/articles/10.3389/frai.2025.1466092/full]
- 56. The two-way relationship between AI and materials science [https://www.nature.com/articles/d42473-025-00107-9]
- 57. Can AI and Robotics Reshape Drug Formulation? [https://pmc.ncbi.nlm.nih.gov/articles/PMC11582498/]
- 58. Artificial Intelligence for Materials Science [https://www.mpie.de/4867299/artificial_intelligence]
- 59. The future of robotics in the pharmaceutical industry. [https://www.essert.com/blog/robotics/future-robotics-pharmaceutical-industry/]
- 60. The Rise of Pharma Robots: Transforming Drug ... [https://www.roboticstomorrow.com/news/2025/02/07/the-rise-of-pharma-robots-transforming-drug-manufacturing-and-research/24079/]
- 61. How pharmaceutical robots are being used in 2025 [https://standardbots.com/blog/pharmaceutical-robots]
- 62. ExoMatter: AI-Powered Materials Research and Development [https://www.exomatter.ai/]
- 63. What is the Materials Science Tetrahedron (Paradigm)? – Materials ... [https://msestudent.com/what-is-materials-science-tetrahedron-paradigm/]
- 64. Metal multi-material additive manufacturing: Overcoming ... [https://www.sciencedirect.com/science/article/pii/S0007850625001489]
- 65. A Systematic Review of Innovative Advances in Multi ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12028848/]
- 66. Accurate predictions on small data with a tabular ... [https://www.nature.com/articles/s41586-024-08328-6]
- 67. About Technology Readiness Levels (TRLs) [https://medicalcountermeasures.gov/trl]
- 68. Integrated Technology Readiness Levels (TRLs) [https://medicalcountermeasures.gov/trl/integrated-trls]
- 69. Technology Readiness Levels applied to Medical Device ... [https://argondigital.com/blog/product-management/technology-readiness-levels-applied-to-medical-device-development/]
- 70. 14 Readiness Level Frameworks: The Guide to TRL, MRL, ... [https://www.itonics-innovation.com/blog/14-readiness-level-frameworks]
- 71. The future of pharmaceuticals: Artificial intelligence in drug ... [https://www.sciencedirect.com/science/article/pii/S2095177925000656]
- 72. Tech, ML and AI: Industry predictions for 2025 [https://www.ddw-online.com/tech-ml-and-ai-industry-predictions-for-2025-33318-202501/]
- 73. Data-driven modeling of process, structure and property in ... [https://www.sciencedirect.com/science/article/abs/pii/S1526612522001529]
- 74. B. STRUCTURAL MATERIALS [https://euromat2025.com/b-structural-materials/]
- 75. Comparison of Additively Manufactured Polymer-Ceramic ... [https://www.mdpi.com/1996-1944/17/1/240]
- 76. Investigation of the Mechanical Properties of a Ceramic ... [https://www.mdpi.com/1996-1944/18/22/5165]
- 77. Innovative fabrication pathways for ultra-high temperature ... [https://www.sciencedirect.com/science/article/pii/S2666539525000847]
- 78. (Ti,Zr,Hf,Ta)CN/SiCN: A new ultrahigh-temperature ceramic ... [https://www.sciopen.com/article/10.26599/JAC.2025.9221104]
- 79. Small data machine learning in materials science [https://www.nature.com/articles/s41524-023-01000-z]
- 80. Materials Science and Engineering | MATSE 81: Materials In Today's World [https://www.e-education.psu.edu/matse81/node/2094]
- 81. Materials science tetrahedron--a useful tool for pharmaceutical research and development - PubMed [https://pubmed.ncbi.nlm.nih.gov/18781623/]
- 82. Materials Informatics 2025-2035: Markets, Strategies, Players [https://www.idtechex.com/en/research-report/materials-informatics-2025/1096]
- 83. Materials science breakthroughs 2025: Trends to watch [https://www.cas.org/resources/cas-insights/materials-science-trends-2025]
- 84. challenges in materials information extraction [https://pubs.rsc.org/en/content/articlelanding/2024/dd/d4dd00032c]
- 85. Extracting accurate materials data from research papers ... [https://www.nature.com/articles/s41467-024-45914-8]
- 86. Top 6 Visualizations for Quantitative Data Analysis Methods [https://chartexpo.com/blog/quantitative-data-analysis-methods]
- 87. Machine Learning-Driven Materials Discovery [https://www.sciencedirect.com/science/article/abs/pii/S235221432500139X]
- 88. Materials Expert-Artificial Intelligence for materials discovery [https://www.nature.com/articles/s43246-025-00928-7]
- 89. Materials Synthesis and Characterization Methods [https://www.nrel.gov/materials-science/materials-synthesis-characterization]
- 90. Synthesis and Processing [https://dmse.mit.edu/research-impact/materials-research-type/synthesis-processing/]
- 91. AI system learns from many types of scientific information ... [https://news.mit.edu/2025/ai-system-learns-many-types-scientific-information-and-runs-experiments-discovering-new-materials-0925]